Dr. PALLAVI JOSHI
Dr.Anand Balasubramaniam, Dr. KAUSHIK MURALI, Mr.Siddhartha Gairola
Abstract
Diagnosis of KCN mandates sophisticated and costly topographers which are limited as diagnostics in institutes, Cost-effective screening tool for diagnosis of earliest KCN is needed.
In a collaborative study with Software professionals, an attempt to create a smartphone-based keratoscope prototype was studied in comparison to keratron based topographer
In this study, we analyzed the steep learning curves with respect :
a.The prototype make: which needed nearly 3 revisions
b. The image quality (mires and optical capture )and fixation target: which also needed prototype modifications and software changes
c.Comparison of images obtained on smartphone-based keratoscope vs standard topographer had an interesting interobserver bias due to heat maps and keartometeric values.
This study highlights the learning curves in a construct of the optical prototype of a Smartphone-based keratoscope which can be used as a screening tool for diagnosis of Keratoconus in the larger population.
Full Text
Diagnosis of KCN mandates sophisticated and costly topographers which are limited as diagnostics in institutes, Cost-effective screening tool for diagnosis of earliest KCN is needed.
Aim: In a collaborative study with Software professionals, an attempt to create a smartphone-based keratoscope prototype was studied in comparison to Keratron based topographer
Setting and design: Developed and validated diagnostic modelling tool(SmartKC)
Methods and material: In this study, we analysed the steep learning curves and applied modifications with
a. The device: In which the prototype evolution was studied
b. The image quality: the effect of app modifications in getting mires and optical capture better
C. The learning in validation of new device and inter observer variability causes.
Results: The protype went through many modifications before the final model was generated. The app included fixation targets, modifications in device distance and tilt and alignments were corrected by repeated discussions and picture captures to get repeatable and comparable images like standard Keratron Topographer.The computation error in SimK once corrected resulted in Overall, SmartKC achieving a sensitivity of 94.1% and specificity of 100.0% as compared to Keratron’s sensitivity of 100.0% and specificity of 64.5% in detecting Keratoconus thus making this as a good option as screening device
Conclusion: This study highlights the learning curves in a construct of the optical prototype of a Smartphone-based keratoscope which can be used as a screening tool for diagnosis of Keratoconus in the larger population.
Key words: Keratoconus (KCN), Smartphone based device, Screening tool
Key message: Collaborated clinical and technical study of the journey of evolving a cost effective screening tool for detecting KCN
Introduction
Keratoconus (KCN) is a progressive, bilateral, ectatic condition of the cornea with a possible inflammatory pathology. It is characterized by progressive thinning and weakening of the stroma, causing it to become conical in shape causing irregular astigmatism and progressive myopia. Advanced stages present as apical scarring, acute hydrops which cause corneal opacities where keratoplasty becomes the only option for treatment. Here comes the need for early detection and treatment to arrest the progression and prevent vision loss.
The prevalence of keratoconus varies among different populations with an estimate of approximately 1/2000 individuals1,2. Recent studies done on the Middle Eastern and Asian population using videokeratoscopy estimate it to be around 0.9% to 3.3%3-10. Particularly in the Indian scenario, Jonas et al, defined keratoconus as a corneal refractive power of more than 48D and estimated the prevalence to be around 2.3%3. Reports of two surveys in the UK indicated a prevalence 4.4 and 7.5 times greater for Asian (Indian, Pakistani, and Bangladeshi) subjects compared with white Caucasians11,12.
Investigative modalities in diagnosing keratoconus ranges from Placido-based Optikon Keratron to tomographers like Oculus Pentacam and Wavelight Oculyzer, which are non-portable and very expensive. This makes early diagnosis inaccessible to large sections of population in middle- and lower-income countries. In addition, with the ongoing COVID-19 pandemic and restrictions on travel, it has become difficult for young children to undergo annual eye examinations at the hospital.
Artificial intelligence has been extensively utilized in the field of medicine in the recent years, which was first proposed by John McCarthy in 195613. Machine learning is a subset of AI, defined as a set of methods that automatically detect patterns in data and then incorporate this information to predict future data14. In ophthalmology, AI has been extensively utilized for diseases like diabetic retinopathy, glaucoma, age related macular degeneration and cataract.
The following article, which could serve as a prototype in developing similar screening devices, explains the journey of making it and is a perfect example to show how a collaboration between medical fraternity and computer science engineers can be helpful in overcoming the hurdles in creating a phone tool to screen such a vision threatening disease with a high prevalence.
Materials and methods
Our study included a collaborated team of software professionals, ophthalmologist and optometrist.The journey of construct of a smart phone-based prototype as a screening tool for keratoconus began with multiple modifications in the tool we named as SmartKC.
The phone tool called SmartKC, comprised of a 3D-printed Placido’s disc attachment, an LED light strip, and an intelligent smartphone app to capture the reflection of the Placido rings on the cornea. The 3 most common designs used for Placido disc attachment are planar, ellipsoid and conical. The ellipsoid variant tends to be very bulky whereas the planar head accommodates rings only to fit the central cornea. Hence, a conical head was found to be the best in terms of being portable yet incorporating around 28 rings as compared to the planar and ellipsoid head. Our final conical Placido head structure has 28 rings, cone length of 70mm, smallest ring (closest to camera) radius of 4mm, largest ring radius of 15mm, and cone vertical angle of 8.93 degrees. Figure 4

Figure 1 : Ellipsoid shape

Figure 2 : Capsule shape and conical shape

Figure 3: Inside conical shape of alternating black and empty rings with supports at 0 and 180 degrees.
To make this a low cost and affordable model a support system for black and empty ring distribution comprised of two 2mm wide supports at 0◦ and 180◦ was chosen. However, these supports resulted in the formation of two V-shaped patterns on the mire image, but the image analysis system interpolated and filled in the missing data points. The outer enclosure also went through a number of design changes with the final one being a capsule shaped enclosure in order to maximise the mire brightness.
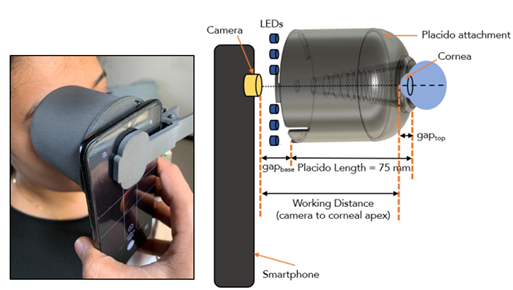
Figure 4 Smart KC
The second challenge was about obtaining proper alignment between principal axis and reference axis as in the ideal set up to obtain good quality mires. If there is an angulation between these 2 axes and is offset by even small amounts like 2mm, the mires produced could be falsely labelled as steep in the area where the Placido is closer to the cornea. To prevent this the phone tool was provided with a red cross hair which had to align with the central mires in order to get good quality images.
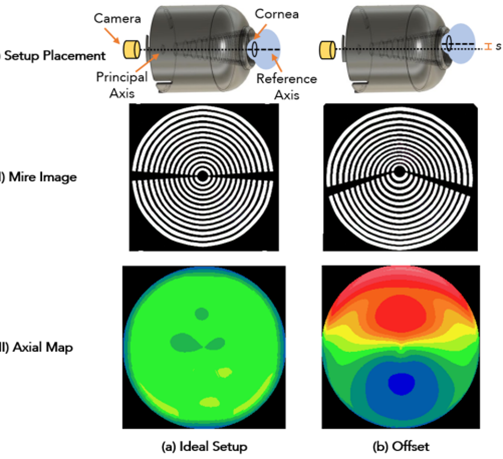
Figure5
False readings can also be obtained when there is an angulation between the 2 axes. To avoid such tilt misalignment and resulting errors, the Placido attachment was modified to have a snug fit around the eye socket (the contour of the orbital cavity), such that the front part of the attachment is fully touching the face. Another important parameter which posed challenge was the Working distance- the distance between the smartphone camera and the corneal apex is another important parameter in getting good quality mires. If it decreases either due to improper positioning or proptosed eyes, it can cause false high readings of corneal curvature.
From the early teething problems and insights, the app was generated for the smart KC to ensure good quality images with good exposure and sharpness with the help of real- time checks using an automatic quality checker module along with the option of auto-capture.
The permutations and combinations in getting the right design was done through a simulation set up, which comprised of a Placido attachment, a diffused light source, a camera with parameters same as that of our data collection smartphone camera, and a reflection surface imitating the cornea. The Placido design and/or reflection surface were varied to obtain mire image as output. Instead of 3D printing each of the design variations during the development of the tool, this kind of simulation set helped us generate quick iterations while maintaining the low-cost budget.
The images were taken by a trained optometrist. On the basis of the location and pixel characteristics of the 3D printed Placido disc and the working distance, an image processor analysed the corneal image, via the arc-step method and Zernike polynomials-based surface fitting, creating the axial and tangential maps. Further, SimK readings and heat maps with the same colour palette as Optikon Keratron (considered as gold standard) was generated so as to be comparable.

Figure 6 : The Process from capture to image generation
Once the prototype was ready, then with institute approval a prospective study to validate the Smart KC against the standard Keratron machine was conducted between June- August 2021 where in a total of 101 eyes were examined-34 with keratoconus and 67 without keratoconus (Normal and with Refractive error). Patients with corneal opacities, scars, nystagmus and severe dry eye were excluded.
The obtained images were then subjected to assessing the specificity and sensitivity as compared to Optikon(Keratron) images by 4 Ophthalmologists were asked to rate 202 pairs of output corneal topography maps, 101 each from SmartKC system and from the gold-standard Keratron device, as keratoconus, non-keratoconus, suspected keratoconus and re-take. Each question comprised of a pair of axial and tangential maps for the same eye, from one of the devices (SmartKC or Keratron). The images were randomized to minimize bias.
It is interesting that we noticed an inter-observer bias prior to refinement of the results. While one of the evaluators diagnosed keratoconus only looking at the pattern of the heat maps, the other evaluator also considered the SimK readings (which was measured initially by the steepest K and flattest K reading in the central 5mm of the cornea) in addition to the heat maps. This initially led to a low specificity of 73.2% but once it was rectified (by computing SimK flattest as the average dioptric power in the flattest meridian in the central 3mm zone, and taking the SimK steepest as the average dioptric power of the meridian 90 degrees opposite to steep) So with correction of wrongly computed SimK the specificity increased by almost 73.2%to 100%.
Table: Inter observer variability due to wrong Sim K computing
| Evaluator | Sensitivity | Specificity |
|---|---|---|
| Dr A (Keratron) | 100% | 65.80% |
| Dr A (Smart KC) | 93.50% | 95.90% |
| Dr B (Keratron) | 100% | 93.30% |
| Dr B (Smart KC) – after correction | 92.30% | 100% |
| Dr B (Smart KC) – before correction | 96.30% | 73.20% |
Results:
With the initial corrections in prototype and image capturing techniques, we were able to overcome the hurdles of validation of obtained topographic maps and the final outcomes showed Overall, SmartKC achieved a sensitivity of 94.1% and specificity of 100.0% as compared to Keratron’s sensitivity of 100.0% and specificity of 64.5%. Specificity means correctly identifying the number of people who don’t have the disease. By having a high specificity, we can reduce the number of patients being falsely labelled as keratoconus which could have a bearing on their mental health.
The SimK1 and SimK2 values computed by the SmartKC closely resembled those of Keratron which is evident with the following results – sim-K1 had Pearson’s r (99) = 0.78, p < 0.01, and sim-K2 had Pearson’s r (99) = 0.62, p < 0.01. the inter-rater agreement computed for the non-retake images across the four doctors in terms of Cohen’s Kappa score was 0.86 for the SmartKC images and 0.98 for the Keratron images. Another important result was that none of the moderate to severe keratoconus cases were missed by the SmartKC tool.
Discussion
There have been a number of smart-phone based portable devices used for the screening of keratoconus developed using the basic principle of projecting Placido mires onto the anterior corneal surface and then capturing those images with a smart-phone which are then processed to
screen for keratoconus15-21. But there have been a lot of limitations which have been overcome in our device. Although the studies describe extensive designing, many of them were never used to screen keratoconus patients18-21. In our study we have been able to generate a successful device and also validated it with a standard machine. Some studies done with smaller sample size
Beling et al obtained a high false positive rate, i.e., even normal individuals were labelled as keratoconus17.Errors in the above studies were mainly due to difficult detection of mires or insufficient coverage of the anterior surface 17,18.
SmartKC also had to undergo a fair share of modifications in structure as well as changes in the functionality. Collaboration with software professionals provided us with a deep insight into the technicalities of this tool which gave us added advantage which helped in evolving a useful device which is cost effective, compact and user friendly. Thus, Smart KC with good specificity, sensitivity and repeatability can be considered as screening device for early detection of Keratoconus in population.
Conclusion:
This study highlights the learning curves in a construct of the optical prototype of a Smartphone-based keratoscope which can be used as a screening tool for diagnosis of Keratoconus, however looking forward is need for larger sampling of Keratoconus patients and use of Artificial intelligence to generate new device specific algorithm’s for increasing the scope of better utility in diagnosis and follow up in keratoconus patients.
References:
- Rabinowitz Y. S. Keratoconus. Survey of Ophthalmology. 1998;42(4):297–319. doi: 10.1016/s0039-6257(97)00119-7.
- Davidson A. E., Hayes S., Hardcastle A. J., Tuft S. J. The pathogenesis of keratoconus. Eye. 2014;28(2):189–195. doi: 10.1038/eye.2013.278.
- Jonas JB, Nangia V, Matin A, et al. Prevalence and associations of keratoconus in rural Maharashtra in central India: The Central India Eye and Medical Study. Am J Ophthalmol. 2009;148(5):760–765.
- Millodot M, Shneor E, Albou S, et al. Prevalence and associated factors of keratoconus in Jerusalem: a cross-sectional study. Ophthalmic Epidemiol. 2011;18(2):91–97.
- Wakend N, Fayad AM, Fadlallah A, et al. Keratoconus screening in Lebanese students’ population. J Fr Ophthalmol. 2012;35(1):23–29.
- Xu L, Wang YX, Guo Y, et al. Prevalence and associations of steep cornea/keratoconus in greater Beijing. The Beijing Eye Study. PLoS ONE. 2012;7(7): e39313.
- Hashemi H, Khabazkhoob M, Fotouhi A. Topographic keratoconus is not rare in an Iranian population: the Tehran eye study. Ophthalmic Epidemiol. 2013;20(6):385–391.
- Hashemi H, Khabazkhoob M, Yazdani N, et al. The prevalence of keratoconus in a young population in Mashad. Iran Ophthalmic Physiol Opt. 2014;34(5):519–527.
- Shneor E, Millodot M, Gordon-Shaag A, et al. Prevalence of keratoconus among young Arab students in Israel. Int J Kerato Ectatic Corneal Dis. 2014;3(1):9–14.
- Hashemi H, Beiranvand A, Khabazkhoob M, et al. Prevalence of keratoconus in a population-based study in Shahroud. Cornea. 2013;32(11):1441–1445.
- Georgiou T, Funnell CL, Cassels-Brown A, O’Connor R. Influence of ethnic origin on the incidence of keratoconus and associated atopic diseases in Asian and white patients. Eye (Lond) 2004; 18:379–83.
- Pearson AR, Soneji B, Sarvananthan N, Sandforth-Smith JH. Does ethnic origin influence the incidence or severity of keratoconus? Eye (Lond) 2000; 14:625–8.
- N. Graham, Artificial Intelligence, vol. 1076, Blue Ridge Summit: Tab Books, Philadelphia, PA, USA, 1979.
- Wei Lu, Yan Tong, Yue Yu, Yiqiao Xing, Changzheng Chen, Yin Shen, “Applications of Artificial Intelligence in Ophthalmology: General Overview”, Journal of Ophthalmology, vol. 2018
- Behnam Askarian, Jo Woon Chong, Fatemehsadat Tabei, and Amin Askarian. 2018. An affordable and easy-to-use diagnostic method fo keratoconus detection using a smartphone. In Medical Imaging 2018: Computer-Aided Diagnosis, Kensaku Mori and Nicholas Petrick (Eds.). SPIE.
- B. Askarian, F. Tabei, G. A. Tipton, and J. W. Chong. 2019. Novel Keratoconus Detection Method Using Smartphone. In 2019 IEEE Healthcare Innovations and Point of Care Technologies, (HI-POCT). 60–62.
- André Beling da Rosa. 2013. An accessible approach for corneal topography. Masters Thesis, Universidade Federal do Rio Grande do Sul.
- Manuel Campos García, Daniel Aguirre-Aguirre, José Antonio Lechuga-Núñez, and Andrés Peña-Conzuelo. 2019. Design of a null-screen for a compact corneal topographer. In Modeling Aspects in Optical Metrology VII, Bernd Bodermann, Karsten Frenner, and Richard M.Silver (Eds.). SPIE
- L V Prasad Eye Institute. 2018. Students Screened for Keratoconus. Retrieved Feb 2, 2021 from https://www.lvpei.org/news-event/studentsscreened-for-keratoconus
- Heliana Bezerra Soares. 2015. Design And Development Of An Ultraportable Corneal Topographer For Smartphones As A Low Cost New Tool For Preventing Blindness Caused By Keratoconus. International Journal of Latest Research in Science and Technology 4 (2015),72–76. Issue 3.
- Sarah Tress. 2018. BullsEYE. Retrieved Feb 2, 2021 https://academy.autodesk.com/portfolios/bullseye



 FP1580 : Learning curve in the construct of smartphone-based keratoscope prototype for KCN screening
FP1580 : Learning curve in the construct of smartphone-based keratoscope prototype for KCN screening
Leave a Comment