Dr. VANITA PATHAK RAY
Semi Final
Abstract
Purpose: To investigate the comparative efficacy and safety of Aurolab Aqueous Drainage Implant(AADI) and the Ahmed Glaucoma Valve(AGV) when implanted in filtration-surgery-naïve secondary glaucoma eyes Methods: Retrospective review of patients with secondary glaucoma who underwent primary tube procedure, either AADI or AGV. Primary outcome measure was IOP and secondary were BCVA, number of antiglaucoma medications(AGMs), complications, failure. Results: 59eyes in AADI with follow-up of 20.3±12.9 months and 67 in AGV with a follow-up of 19.9±18.2 months, were included. Preoperative IOP, AGM, and BCVA did not differ but at last visit, IOP(p=0.005) and AGM(P<0.001) and failure rate (p=.047)was significantly lower in the AADI group. Complication rates were comparable. Conclusions: Both procedures were effective in reduction of IOP and need for AGM but was significantly so in AADI along with rate of failure and this affordable GDD could have a tremendous impact in our country.
Full Text
Short title: Primary implantation in secondary glaucoma: AADI vs AGV
Author/s: Vanita Pathak-Ray FRCS (Ed), FRCOphth (Lon) Centre for Sight, Road No 2 Banjara Hills, Hyderabad, India 500034
Orcid ID no for Dr. Vanita Pathak-Ray: 0000-0002-7830-283X
Corresponding author:
Dr. V Pathak-Ray, FRCS (Ed) FRCOphth (Lon)
Director Glaucoma, Centre for Sight, Road No 2 Banjara Hills, Hyderabad, India 500034
Email: vpathakray@gmail.com
Cell: +91 9000420031
Funding: Nil
Conflict of Interest: Nil relevant (Santen, Alcon/Novartis, Allergan)
Tables: 6
Figures: 4
Primary implantation of Glaucoma Drainage Device in Secondary Glaucoma: comparison of AADI vs AGV
Abstract
Purpose
To investigate the comparative efficacy and safety of Aurolab Aqueous Drainage Implant (AADI) and the Ahmed Glaucoma Valve (AGV) when implanted in filtration-surgery-naïve secondary glaucoma eyes
Methods:
Retrospective review of patients with secondary glaucoma who underwent primary tube procedure, either AADI or AGV. Primary outcome measure was IOP and secondary were BCVA, number of antiglaucoma medications (AGMs),
complications.
Results
59eyes in AADI with follow-up of 20.3±12.9 months and 67 in AGV with a follow-up of 19.9±18.2 months, were included. Preoperative IOP, AGM, and BCVA did not differ but at last visit, IOP and AGM was significantly lower and
complete success significantly higher in the AADI group (all p<.001). Serious complication rates were comparable
Conclusions:
Both procedures were effective in reduction of IOP and need for AGM, but this was significantly so in the AADI group along with highere rate of complete success and therefore this affordable GDD could have a tremendous impact in
our country.
Key words :
Glaucoma Drainage Device, GDD, Valved GDD, non-valved GDD, tubes, hypertensive phase
Introduction:
Secondary glaucomas form a significant proportion of all glaucomas and the majority tend to be very refractory to treatment. Such eyes generally tend to have more inflammation (uveitic glaucoma, neovascular glaucoma, silicone
oil induced glaucoma etc.) or tend to have membranes growing over the angle to shut it down (neovascular glaucoma, Irido-corneal endothelial syndromes etc.) or tend to have scarred conjunctiva due to previous surgeries (post
vitreo-retinal surgeries, post keratoprosthesis etc.) or may have a combination of one or more of these factors.
Not only are they poor responders to medical management but quite often fail after trabeculectomy (trab) as well, despite use of adjuvants. Thus, it is not unusual for these eyes to undergo multiple trabs before a glaucoma
drainage device (GDD) is considered or the patient referred on as the technical skill may not be available locally. Several authors have reported that when such eyes of refractory glaucomas with failed trab/s undergo a GDD, then
it too tends to fail. [1-4] Therefore a therapeutic approach that may result in better outcomes in eyes that have refractory secondary glaucoma is to implant GDDs primarily.
The incidence of secondary glaucomas in India has been reported to be at a variable rate – 6% [5] to 21.8%.[6] Although the rate may be unequable, most authors agree that such glaucomas tend to be very refractory to treatment
and a sizeable number of such eyes end up with GDD surgery. The two types of GDD surgery available in India are the Ahmed Glaucoma Valve (AGV, New World Medical Inc., Rancho Cucamonga, California, USA) and the Aurolab Aqueous
Drainage Implant (AADI, Aurolabs, India).
The AGV is a valved device with a flow restrictor which theoretically remains closed at low intraocular pressure (IOP) and is thus considered to be protective for hypotony. The AADI is an indigenously manufactured, inexpensive,
non-valved device, the design inspiration of which is the Baerveldt Glaucoma Device (BGD, Advanced Medical Optics, Santa Ana, California, USA) – 350 mm2 model.
Although there are a few studies that have compared AGV and AADI in refractory glaucomas in adults, [7-9] none of these have reported the outcomes of primary implantation of GDD in secondary glaucomas. The objective of this
study was to investigate the comparative efficacy and safety of AADI and AGV when implanted in filtration-surgery-naïve secondary glaucoma eyes.
METHODS :
Study design:
This was a retrospective, comparative, interventional study wherein a review of charts of consecutive adult patients who underwent GDD surgery and were followed-up between January 2017 to June 2021 by a single fellowship-trained
surgeon and who had at least 3 months of documented post-op follow-up, was undertaken. Ethical clearance was obtained from an Independent Ethics Committee; the study adhered to the principles as laid down by the Declaration of
Helsinki. Informed written consent for surgery was obtained from all the eligible participants.
Inclusion criteria:
Consecutive AGV/AADI surgery with a minimum of 3 months follow-up.
Exclusion criteria:
GDD in those eyes that underwent prior trabeculectomy were excluded and so were those eyes where Goldmann Applanation Tonometry (GAT) was either not possible or compliance was poor (e.g. Kerato-prosthesis, pediatric eyes etc).
Initially a detailed history was obtained from all patients and the eyes underwent a comprehensive examination. This included – best-corrected visual acuity (BCVA) assessment with a Snellen chart, detailed slit-lamp
biomicroscopy, intraocular pressure (IOP) assessment with Goldmann applanation tonometry (Zeiss SL 130 slit lamp with Goldmann style applanation tonometer AT 030), gonioscopy with Sussman 4-mirror, dilated stereo fundoscopy with
+66D Volk lens (Volk Instruments, OH, USA).
Visual fields, where possible, was documented by the Humphrey field analyser (HFA, Carl Zeiss Meditec, Jena, Germany). Follow-up visits were scheduled as per clinical indication but for the purpose of the study, data was
documented at day 1, week 1, week 6, month 3, month 6 and year 1 and at last follow-up postoperatively.
Surgical methods:
Surgical procedure of AADI has been described in detail previously. [8,10] All AADI plates were placed under adjacent recti and none of the plates were trimmed. For AGV standard implantation between muscles was followed. Under
peribulbar block and sterile conditions, a fornix-based conjunctival opening was created, commonly in the supero-temporal (ST) quadrant. The implant was primed and then anchored to the sclera, 10 mm posterior to the the limbus,
with a long pass of a preplaced 6-0 vicryl suture (Braided coated polyglactin 910 violet; Ethicon, Johnson & Johnson, HP, India).
The needle was then threaded through the eyelets on the endplate and it was inserted into the ‘pocket’, positioned between muscles and secured with a knot which was rotated into the fixation eyelet. The tube length was shortened
to approximately 3 mm with a bevelled tip opening toward the cornea. A 23-gauge needle was used to create a track 2mm behind the limbus through which the tube was inserted into the anterior chamber just anterior and parallel to
the iris for anterior chamber placement and behind the iris for a sulcus placement.
The tube was inserted through the needle track and secured to the sclera with a figure-of-eight 10-0 nylon suture (monofilament polyamide black, Ethilon; Ethicon, Johnson & Johnson, Himachal Pradesh, India). For both the types
of tubes, almost the entire length of the tube was covered with a corneal patch graft, prepared a-priori,[10] or a scleral patch graft. Patch graft was secured over the tube with either fibrin glue or 10-0 nylon suture. The
conjunctiva and tenon were then brought forward and secured back into position with 8-0 vicryl (Braided coated polyglactin 910 violet; Ethicon, Johnson & Johnson, HP, India) wing and continuous sutures.
After the procedure, a subconjunctival injection of steroid (dexamethasone 4 mg) was given. The eye was patched and removed 24-hrs later. Post-operatively topical antibiotics moxifloxacin was used four times daily for one week
and topical cycloplegic eye drops (Homatropine 2%) was used as per requirement for 1-2 weeks. Topical steroid drops (difluoro prednisolone butyrate acetate, or DFBA 0.05%) was used 2-hourly for 1 week and thereby tapered slowly
over 8 to 12 weeks.
Most significantly, AGM was recommenced in the AGV group in the early post-operative period as soon as IOP>14 mmHg. The AGM commenced were mostly topical aqueous suppressants – beta blockers, carbonic anhydrase inhibitors or
alpha-adrenergic agents, either alone or in fixed drug combinations. This was not the therapeutic approach in AADI. Outcome criteria: Primary outcome measure was IOP and secondary outcome measures were the number of
anti-glaucoma medication (AGM), LogMAR best corrected visual acuity (BCVA) and complications.
Complete success was defined as an IOP ≥ 5 mmHg and ≤ 21 mm Hg. When the above IOP criteria was met with AGM, it was considered as qualified success. Failure was defined as the inability to meet IOP criteria, loss of perception
of light, explantation of device or any additional glaucoma surgery to reduce IOP. Any eye which underwent a re-procedure or which had reduction in visual acuity (VA) of 2 lines or more was considered as a serious complication.
Hypertensive phase was defined by a tense cystic bleb around the plate with much increased height accompanied with IOP ≥21 mmHg with or without AGM, after reduction of IOP to less than 22 mmHg during the early postoperative
period and not caused by tube obstruction, from the third week onwards for AGV and post-suture autolysis after the sixth week onwards for AADI.
Statistics:
Descriptive statistics was performed to compare baseline demographic and ocular characteristics of the treatment groups. Descriptive data is presented as Mean±Standard Deviation. Normality was checked via the Shapiro-Wilk test.
Accordingly, univariate comparisons were performed using the paired t test or Wilcoxon-Signed rank test within group and Independent t test or Mann-Whitney U test for between-group comparisons. Chi-squared test or Fisher’s exact
test was used for categorical variables. Snellen visual acuity was converted to logarithm of minimal angle of resolution (logMAR) for analysis.
IOP and AGM data was censored if explanted or second glaucoma surgery was needed; visual acuity data was not censored. Survival analysis by Kaplan-Meier and risk factors for treatment failure were assessed for statistical
significance by Cox proportional hazard. All statistical tests were 2-sided, and statistical significance was defined as p<0.05. Statistical analyses were performed using the statistical software Stata 12.1 (StataCorp, College
Station, TX).
Results:
A total of 126 eyes of 119 subjects underwent primary GDD in secondary glaucoma. All the surgeries were performed by a single senior fellowship-trained glaucoma specialist. 59 eyes underwent AADI, and 67 eyes underwent AGV. All
the eyes in the AADI group had a follow-up of more than 3 months and were included; 6 of 67 eyes in the AGV had less than 3 months follow-up and were excluded. Mean follow-up was 20.3±12.9 months in the AADI group and 19.8±11.8
months in the AGV group. Supero-temporal (ST) was the most common quadrant for placement of the GDD in both the groups – 98.4% (n=60) in the AGV group and 91.5% (n=54) in the AADI group.
All the rest in both the groups were placed in the infero-temporal (IT) quadrant. 50.8% (n=31) AGV tubes and 40.7% (n=24) AADI tubes were placed in the ciliary sulcus (CS). 11.8% (n=7) eyes underwent simultaneous GDD and
cataract surgery in the AADI group and 6.5% (n=4) in the AGV group. An additional 4.9% eyes (n=3) underwent simultaneous flanged technique of sclera fixated intra-ocular lens implant (SF-IOL). Retinal diseases and surgery
related to it formed the largest etiological category of secondary glaucoma in both the groups. (Table 1)
Table 1: Etiology of secondary glaucoma between the two groups – Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV)
| AADI n=59 | AGV n=61 | p | |
| Neovascular glaucoma | 12 | 25 | 0.055 |
| Post Retinal surgery | 13 | 19 | 0.177 |
| Aphakic/ pseudophakic | 6 | 11 | 0.423 |
| Post Corneal Transplant (PK/ DSEK) | 7 | 1 | 0.024 |
| Uveitis | 10 | 2 | 0.012 |
| Others | 11 | 3 | 0.019 |
Both groups were well matched in terms of baseline characteristics. (Table 2)
Table 2: Baseline characteristics between the two groups – Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV)
| AADI n=59 | AGV n=61 | p | |
| Age in years | 47.2 ± 19.7 | 45.9 ± 17.0 | 0.689 |
| Intraocular Pressure in mmHg Mean ± SD | 35.8 ± 10.6 | 33.1 ± 11.7 | 0.139 |
| Anti-glaucoma medications (number) Mean ± SD | 3.8 ± 0.9 | 4.1 ± 0.7 | 0.542 |
| BCVA (LogMAR)Mean ± SD | 1.1 ± 0.7 | 1.2 ± 0.7 | 0.363 |
Intraocular pressure and anti-glaucoma medications: Table 3 and 4, Figure 1 and 2
Table 3: Outcomes in Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV) groups
| AADI | AGV | p | |
| Intraocular Pressure in mmHg Mean ± SD | 12.9 ± 3.7 | 15.7 ± 2.7 | <0.001 |
| Anti-glaucoma medications (number) Mean ± SD | 0.6 ± 0.9 | 1.8 ± 1.1 | < 0.001 |
| BCVA (LogMAR) | 1.0 ± 0.7 | 1.0 ± 0.7 | 0.865 |
| Complete success (n%) | n=34 (57.6%) | n=9 (14.7%) | <0.001 |
| Total success (n%) | n=54 (91.5%) | n=49 (80.3%) | 0.078 |
Table 4: Baseline and follow-up intraocular pressures (IOP) and anti-glaucoma medications (AGM) for the two groups, Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV)
| fsfd | AADI IOP | AGV IOP | p value | AADI | AGV number of AGM | p value |
| Mean ± SD | Mean ± SD | |||||
| Pre-op | 35.8 ± 10.6 | 34.8 ± 13.3 | 0.139 | 3.8 ± 0.9 | 4.1 ± 0.7 | 0.542 |
| †POD1 | 22.3 ± 11.7 | 12.1 ± 5.2 | <0.001 | – | – | – |
| ‡POW1 | 20.6 ± 9.6 | 14.0 ± 7.0 | <0.001 | 1.7 ± 1.2 | 0.4 ± 0.7 | <0.001 |
| POW6 | 9.3 ± 7.6 | 16.9 ± 6.5 | <0.001 | 0.8 ± 1.4 | 1.5 ± 1.0 | 0.002 |
| §POM3 | 13.1 ± 4.2 | 16.0 ± 5.0 | 0.008 | 0.6 ± 0.9 | 1.8 ± 1.1 | <0.001 |
| POM6 | 11.9 ± 3.7 | 14.8 ± 3.6 | <0.001 | 0.7 ± 1.1 | 1.7 ± 1.0 | <0.001 |
| Last follow-up | 12.9 ± 3.7 | 15.7 ± 2.7 | <0.001 | 0.6 ± 0.9 | 1.8 ± 1.0 | <0.001 |
| †POD | ‡POW | §POM | ||||
| Post-op Day | Post-op Week | Post-op month |
Figure 1. Intraocular pressure (IOP) in Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV) groups
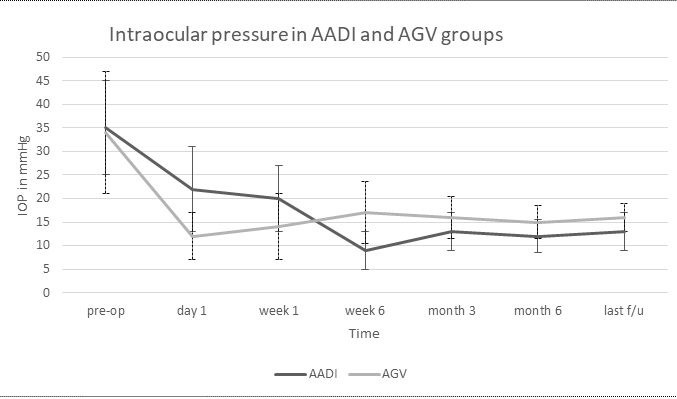
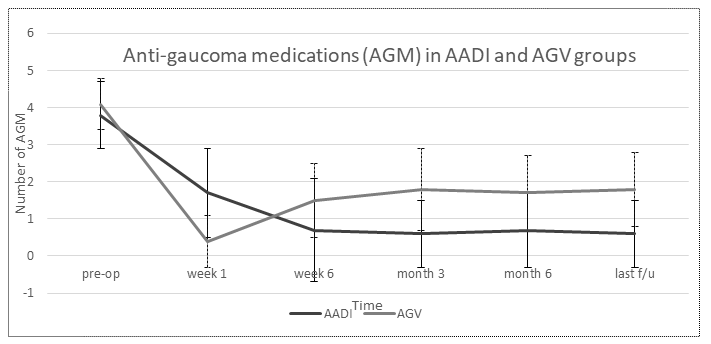
Figure 2. Number of anti-glaucoma medication/s (AGM) in Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV) groups
In the AADI group, mean IOP decreased from 35.8 ± 10.6 mmHg at baseline to 12.9 ± 3.7 mmHg at last follow-up (p<0.001); the percentage reduction was 64%. Similarly, in the AGV group, mean IOP decreased from 34.8 ± 13.3 mmHg at baseline to 15.7 ± 2.7mmHg at last follow-up (p<0.001) and the percentage reduction was 54.8%.
The AGV group had a significantly lower mean IOP than the AADI group at the day 1 and the week 1 follow-up visit (both p <0.001). However, the mean IOP in the AADI group was significantly lower than the AGV group at every post-operative visit thereafter Table 4.
Similarly, there was a significant reduction in the need for medical therapy in both treatment groups (p<0.001) with a significantly lower need for AGM in the AADI group at every post-operative visit after the week 1 visit. At the final follow-up visit, the percentage reduction in AGM was 84% in the AADI group and 56% in the AGV group. Figures 1 and 2 plot the IOP and AGM between groups at various timepoints.
Complications: Table 5 enlists all the complications in each group. Notably the early complications and the total number of complications in the AGV group were significantly more than the AADI group, but late as well as serious
complications, as defined previously, did not differ between groups. Table 6
Table 5: Early and late complications for the two groups, Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV)
| Early complications ≤3 months | AADI (n) | AGV (n) | p |
| Hyphaema | 2 | 8 | – |
| Hypotony / Choroidals | 4 | 5 | – |
| Shallow AC / aqueous misdirection | – | 2 | – |
| Fibrin / Hypopyon | 3 | 5 | – |
| Endophthalmitis | – | 1 | – |
| Conj gape / retraction | 6 | 5 | – |
| Tube block | 1 | 2 | – |
| Tube / Plate exposure | 2 | 3 | – |
| Vitreous Haem | 1 | 2 | – |
| Total early complications ≤ 3 months | 19 (32.2%) | 33 (54%) | 0.015 |
| Late complications > 3 months | AADI (n) | AGV (n) | – |
| Corneal Decompensation | 2 | 2 | – |
| Tube block | 1 | – | – |
| Patch graft melt | – | 1 | – |
| Vitreous Haem | – | 2 | – |
| Endophthalmitis | – | 1 | – |
| Spontaneous extrusion | – | 1 | – |
| Tube-endo touch | 1 | – | – |
| Total late complications > 3 months | 4 (6.7%) | 7 (11.4%) | 0.373 |
| Early and Late complications | 23 (38.9%) | 40 (65.5%) | 0.003 |
Table 6: Interventions for all complications and serious complications for the two groups, Aurolab Aqueous Drainage Implant (AADI) and Ahmed Glaucoma Valve (AGV)
| Interventions for all complications | AADI (n) | AGV (n) | p |
| Suturelysis in OR | 1 | – | – |
| Conj suturing / CLAG | 2 | 4 | – |
| Repeat Patch graft | – | 1 | – |
| CD drainage | 2 | 2 | – |
| Tube trimming / repositioning | 2 | 2 | – |
| Vitrectomy (Pars plana, anterior, IZHV) | 1 | 3 | – |
| RD surgery | – | 1 | – |
| AC wash / reformation | 1 | 2 | – |
| Total interventions | 9 (15.2%) | 15 (24.5%) | 0.201 |
| Serious Complications(Interventions + VA worse ³ 2lines or NLP) | 13 (22%) | 19 (31.1%) | 0.78 |
Visual acuity: Almost a similar proportion of eyes in both groups (n=47, 79.7% in the AADI group and n=49, 80.3% in the AGV group; p=0.927) had either no change or an improvement in BCVA. Overall, BCVA improved
marginally in both the groups, but this was not statistically significant (p=0.865) One eye in the AGV group lost perception of light but none in the AADI group did so. Four eyes in the latter had vision worse by more than 2
lines, whereas this occurred in 2 eyes in the former group.
Hypertensive phase: HTP was seen less frequently in the AADI group (n=14, 23.7%) and much more frequently in the AGV group (n=29, 50.8%, p=.006) where it was detected most commonly between 3-6 weeks, despite
the use of early aqueous suppressants.
Outcomes: Complete success was seen in 34 eyes (57.6%) in AADI group and another 20 eyes (33.9%) achieved IOP control with medication (qualified success). Therefore, total success in the AADI group was 54 eyes (91.5%). Complete
success was seen in 9 eyes (14.7%) in the AGV group and an additional 40 eyes (65.6%) as qualified success (total success 49 eyes, 80.3%). Thus, even though the overall success was not significantly higher in the AADI group at
last follow-up (p=.078), the complete success achieved in the AADI group was statistically significant (p<.001).
A total of 5 eyes failed in the AADI group (8.4%) and 11 eyes (18%) did so in the AGV group (p =.123). Device explantation was commoner in AADI (n=4) whereas failure on IOP criterion and the need for further glaucoma surgery was
higher in the AGV group (n=8). One eye in the AGV group failed on VA criterion, but none did so in the AADI group. A Kaplan Meier plotting of cumulative probability of complete success was 66%, 60% and 50% in the AADI group at
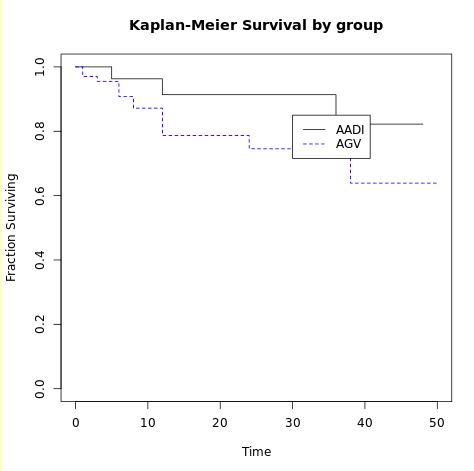
year 1, 2 and 3 respectively whereas it was 22%, 18% and 15% in the AGV group (log rank <.001). The total success was 92%, 90% and 82% at year 1, 2 and 3 respectively in the AADI group and that in the AGV group was 88%, 72% and
62%. Figures 3 and 4 show the Kaplan Meier survival by group for complete and total success.
Figure 3. Kaplan Meier survival by group – complete success
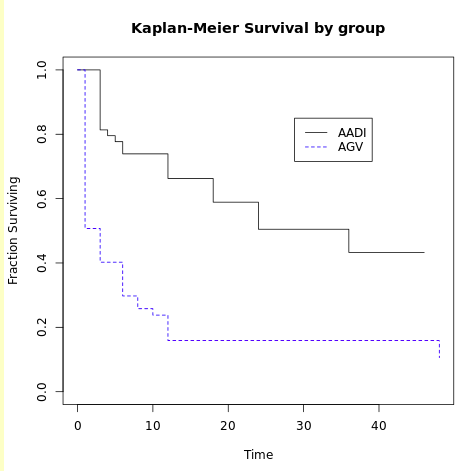
Figure 4. Kaplan Meier survival by group – total success
Cox proportional hazards model was constructed to determine baseline, pre-operative, intra-operative and post-operative characteristics for failure. Hypertensive phase and AGV were found to be significant in multivariate
analysis.
Discussion
This retrospective study compared the primary implantation of non-valved GDD (AADI) and the valved GDD (AGV) in secondary glaucomas which were filtration surgery naïve, and demonstrated that non-valved GDD can achieve
significantly lower IOP with much lesser number of AGM when compared to valved AGV. Furthermore, even though the overall success rate did not differ between groups, the complete success rate was higher in the AADI group.
Similarly, though the incidence of serious complications did not differ, the early and total complications occurred at a significantly increased rate in the AGV group.
Most studies including the large, randomised control trials comparing valved vs non-valved GDD viz. the Ahmed vs Baerveldt study (AVB study)[11] or the Ahmed Baerveldt Comparison study (ABC study)[12] have reported on mixed
cohorts of primary and secondary glaucomas, a significant proportion of which had undergone previous trabeculectomy. However, the ABC study did stratify its cohort into four groups and demonstrated that the best results were
obtained in the group of primary glaucomas which had undergone previous intra-ocular surgery and the worst outcomes were seen in the neovascular group.
The ABC study reported that the AGV group failed on IOP end points whereas the Baerveldt Glaucoma Implant (BGI) failed on safety endpoints. Pooled data from both the studies,[13] showed that the failure rate was 37% in the BGI
group vs 49% in the AGV group at 5 years but both demonstrated that lower target IOP is achieved in the non-valved BGI group.
The AADI is very similar to the BGI 350 and in a study by Puthuran and colleagues reporting the intermediate outcomes[14] in AADI, the failure rate was 9.5% at year 1, 27.8% at 2 years, 38.9% at 3 years and 50.1% at 4 years. The
failure rate reported by Puthuran et al at year 2 is much higher than that reported previously by Pathak Ray and colleague (13%)[15] as well as in this study too (10%). Puthuran et al have reported increased failure rate in
primary (vs secondary) glaucomas perhaps due to the higher rate of previous filtration surgery, providing credence to the hypothesis that previous filtration surgery may be implicated in lower success rates in GDD surgery, as
noted by several other authors too.[1-4]
Results in this study are similar to the pooled data from the ABC and AVB studies at the end of 5 years[13] which showed that non-valved surgery group produced a lower mean IOP on fewer medications, had lower failure rates than
the AGV group. However, the BGI group carried a risk of hypotony which was not seen in this current study.
There are very few studies originating in India which have compared the non-valved Aurolab Aqueous Drainage Implant (AADI), with the valved AGV. We have previously reported the outcomes of the two in a mixed cohort of primary
and secondary glaucomas at 1 year – 92.3% overall success in the AADI group and 80.5% was seen in the AGV group (p<.001).[16] Pandav et al,[17] also reported in a mixed cohort - overall success was 73.08% and 58.18% in the AADI
and AGV groups respectively at 3 years. The corresponding overall success rates in the current study are 92% and 88% at 1 year and 82% and 62% at 3 years in the AADI and AGV groups respectively.
Where intra-ocular positioning of the tubes of either of the GDD was concerned, the first choice in pseudophakic and aphakic eyes (and in all those that had simultaneous cataract extraction) was in the CS.[18] Notably, 2
consecutive pseudophakic neovascular glaucoma eyes, with AC placement of tubes, showed progressive shallowing of the AC with progression of ectropion uvaeae, had to be re-sited in the CS. Following this set-back, the practice of
CS placement was routinely adopted in all pseudophakes, aphakes and those combined with cataract surgery. However, in eyes that undergo simultaneous SFIOL, it may be better to place the tube in the AC rather than risk IOL tilt
and subluxation and this is the authors preferred technique.[19]
Placement of the tube in CS has several advantages – it helps to minimise tube-endothelial touch, endothelial cell loss,[20] subsequent corneal decompensation, and interventions related to it – tube trimming, tube repositioning,
tube extender, corneal graft etc. The rate of corneal decompensation was low in this study and was less than 5%. The rate of corneal decompensation in a recent large retrospective cohort[21] of over 1600 eyes with GDD
implantation was 5%, but it has been reported to be as high as 17%[22] and 19%.[23] Beatson et al[21] found age, post-operative hypotony, tube-cornea touch, Fuchs and Irido-corneal endothelial (ICE) syndrome to be significant
risk factors.
The preferred option in ICE syndrome should be the CS as far as feasible which may help to postpone keratoplasty for a significant period, if not avoid it altogether, as notified in a previous case series.[24] Tube-cornea touch
and subsequent adverse events can be avoided with a CS placement or even a placement in the posterior segment; however, the latter requires pars plana vitrectomy (PPV). None of the GDD surgeries in the current study were
combined with pars plana vitrectomy (PPV) nor were any tubes placed in the posterior segment.
Another very important aspect of GDD implantation is the occurrence of the hypertensive phase. It tends to occur early and with greater severity in the valved devices as it is hypothesized that the early access of the aqueous to
the nascent bleb around the endplate in the early post-operative period allows pro-inflammatory cytokines and other ligands[25] to set up a fibrotic response in the subconjunctival space. It is postulated that this thickens and
forms encystment of the bleb lining leading to increased resistance and thus and increase in IOP occurs.
The incidence of HTP in AGV has been reported variably in literature –Ishida and colleague reported 26% and 35% respectively in a White and African-American cohort,[26] 33% was reported by Abe et al[27] in a mixed racial cohort,
31.3% was reported in a Korean population,[28] 41.1% in an Egyptian population,[29] 58.3%[16] 41.8% [17] and 63.93%[30] in three different studies in the Indian population. On the other hand, HTP occurs rather more infrequently
and presents much later in AADI surgery, only after the occlusive suture autolyses, and has been reported to occur in approximately 20% of eyes.[10] The incidence of HTP in the AADI group in this study was comparable.
In the pooled analysis of the AVB and ABC studies[13] AGV had a significantly higher rate of HTP (60%) when compared to BGI (20%). Therefore, the results obtained in the current study are consistent and comparable with the rate
of HTP in the valved as well as the non-valved GDD as reported in literature. A recent strategy to minimise the hypertensive phase in AGV is the use of early aqueous suppression.[31] This therapeutic intervention was adopted
uniformly in the AGV group and aqueous suppressants were commenced as soon as the IOP was detected in the early teens. This has resulted only in a marginal improvement in the HTP rate in the AGV group in this study (50.8%)
compared to previously reported (58.3%).[16] Predictably therefore it is this phase as well as the use of AGV implant itself that was found to be significant factors for failure.
Even though complications occurred more frequently in the AGV group (65.5% vs 38.9%), most of these in both the groups were transient and were managed conservatively. These rates are comparable to those reported by Pandav et
al[17] in the AGV group (62.9%) but not in the AADI group (75.2%). Most notably, none of the eyes in this study had persistent hypotony related complications in the AADI group whereas Pandav et al have reported a rate of 3.5% in
the AADI group and 0.5% in the AGV group. This difference in the AADI group between the 2 studies may be explicable by the fact that all the surgeries in this series were performed by a single surgeon.
Though there is not much variability in the surgical technique for the performance of an AGV, considerable technical skill is required in the implantation of a non-valved implant and may differ from surgeon-to-surgeon based on
their experience. However, the interventions required for some of these complications are rather more comparable between the groups in both the studies – 24.5% in AGV vs 17% reported by Pandav et al[17] and 15.2% in the AADI
group vs 16% reported by the same authors.[17] Level and years of experience of the surgeon can reduce complications and interventions for these. This is perhaps reflected in AADI implantation also – in a previous report by
Pathak Ray and colleague, [8] rate of interventions was 30.7% (vs 15.2% in this study) whereas it remained comparable in AGV implantation in both the studies.
It may also be indicative of a reduced rate of interventions in primary implantation of non-valved surgery in secondary glaucomas. Nonetheless, this hypothesis can be proven only when a study is conducted inspecting primary and
secondary implantation of AADI in secondary glaucomas. Visual acuity was comparable between the groups, but 4 eyes had vision worse than >2 lines in the AADI group and 2 eyes in the AGV group; this difference was not
significant. Only one eye developed no light perception (following endophthalmitis) in the entire cohort and this was in the AGV group.
The retrospective nature of the study meant that inevitably there was a selection bias, evident by the somewhat mismatched pre-operative etiological characteristics between groups; however pre-operative IOP and AGM did not
differ. This limitation was partially overcome by the inclusion of consecutive eligible eyes with the surgery being performed by a single surgeon in both the groups, eliminating variability of surgical technique as a confounding
factor. Another perceived limitation is the patchy availability of endothelial cell count pre-and-post operatively; therefore, this parameter could not be analysed in this study.
In conclusion both devices are efficacious and safe when implanted primarily in filtration-surgery-naïve eyes with secondary glaucoma. Nevertheless, IOP in AGV, the costlier of the two devices, is more likely to be controlled by
AGM (with lifelong implication of cost), more likely to fail with greater incidence of hypertensive phase when compared to AADI. On the other hand, AADI implant showed a higher rate of complete success with significantly lower
IOP and requirement of fewer AGM with less frequent complications than AGV implant.
References:
1. Sungur G, Yakin M, Eksioglu U, Satana B, Ornek F. Assessment of conditions affecting surgical success of Ahmed glaucoma valve implants in glaucoma secondary to different uveitis etiologies in adults. Eye (Lond). 2017
Oct;31(10):1435-1442.
2. Zhu Y, Wei Y, Yang X, Deng S, Li Z, Li F, et al. Clinical Outcomes of FP-7/8 Ahmed Glaucoma Valves in the Management of Refractory Glaucoma in the Mainland Chinese Population. 2015 PLoS ONE 10(5): e0127658.
3. Souza C, Tran DH, Loman J, Law SK, Coleman AL, Caprioli J. Long-term outcomes of Ahmed glaucoma valve implantation in refractory glaucomas. Am J Ophthalmol, 2007. 144(6): p. 893–900. pmid:17916318
4. Djodeyre MR, Peralta Calvo J, Abelairas Gomez J. Clinical evaluation and risk factors of time to failure of Ahmed Glaucoma Valve implant in pediatric patients. Ophthalmology. 2001 Mar;108(3):614-20.
5. Krishnadas R, Ramakrishnan R, Secondary Glaucomas: The Tasks Ahead Community Eye Health. 2001; 14(39): 40–42.
6. Gadiia R, Sihota R, Tanuj Dada, Viney Gupta, MCurrent profile of secondary glaucomas Indian J Ophthalmol. 2008 Jul-Aug; 56(4): 285–289.
7. Pandav SS, Seth NG, Thattaruthody F, Kaur M, Akella M, Vats A, Kaushik S, Raj S. Long-term outcome of low-cost glaucoma drainage device (Aurolab aqueous drainage implant) compared with Ahmed glaucoma valve. Br J Ophthalmol.
2020 Apr;104(4):557-562.
8. Pathak Ray V, Rao DP. Surgical outcomes of a new affordable non-valved glaucoma drainage device and Ahmed glaucoma valve: comparison in the first year. British Journal of Ophthalmology 2019;103:659-665.
9. Rathi SG, Seth NG, Kaur S, Thattaruthody F, Kaushik S, Raj S, Pandav SS, Ram J. A prospective randomized controlled study of Aurolab aqueous drainage implant versus Ahmed glaucoma valve in refractory glaucoma: A pilot study.
Indian J Ophthalmol. 2018 Nov;66(11):1580-1585.
10. Pathak Ray V, Rao DP. Surgical Outcomes of a New Low-Cost Nonvalved Glaucoma Drainage Device in Refractory Glaucoma: Results at 1 Year. J Glaucoma. 2018;27(5):433-9.
11. Christakis PG, Kalenak JW, Tsai JC, Zurakowski D, Kammer JA, Harasymowycz PJ, Mura JJ, Cantor LB, Ahmed II. The Ahmed Versus Baerveldt Study: Five-Year Treatment Outcomes. Ophthalmology. 2016 Oct;123(10):2093-102
12. Budenz DL, Barton K, Gedde SJ, Feuer WJ, Schiffman J, Costa VP, Godfrey DG, Buys YM; Ahmed Baerveldt Comparison Study Group. Five-year treatment outcomes in the Ahmed Baerveldt comparison study. Ophthalmology. 2015
Feb;122(2):308-16.
13. Christakis PG, Zhang D, Budenz DL, Barton K, Tsai JC, Ahmed, II, et al. Five-Year Pooled Data Analysis of the Ahmed Baerveldt Comparison Study and the Ahmed Versus Baerveldt Study. Am J Ophthalmol. 2017;176:118-26.
14. Puthuran GV, Palmberg P, Wijesinghe HK, Krishnadas SR, Robin A. Intermediate-Term Outcomes of an Affordable Aqueous Drainage Implant in Adults with Refractory Glaucoma. Ophthalmol Glaucoma. 2019 Jul-Aug;2(4):258-266.
15. Ray VP, Rao DP. Two-year Outcomes of the New Low-cost Non-valved Aurolab Aqueous Drainage Implant in Refractory Glaucoma. J Glaucoma. 2020 Sep;29(9):767-772.
16. Pathak Ray V, Rao DP. Surgical outcomes of a new affordable non-valved glaucoma drainage device and Ahmed glaucoma valve: comparison in the first year. British Journal of Ophthalmology 2019;103:659-665.
17. Pandav SS, Seth NG, Thattaruthody F, Kaur M, Akella M, Vats A, et al. Long-term outcome of low-cost glaucoma drainage device (Aurolab aqueous drainage implant) compared with Ahmed glaucoma valve. Br J Ophthalmol.
2020;104(4):557-62.
18. Pathak Ray V. Placement of the tube of a glaucoma drainage device: different horses for different courses. Indian J Ophthalmol. 2022. Doi 10.4103/ijo.IJO_2877_21
19. Pathak-Ray V, Bansal AK, Malhotra V. Combining flanged intrascleral IOL fixation with Glaucoma Surgery: Initial experience. Eur J Ophthalmol. 2021 Dec 13:11206721211066390. doi: 10.1177/11206721211066390. ePub ahead of print
20. Tan AN, Webers CA, Berendschot TT, de Brabander J, de Witte PM, Nuijts RM, Schouten JS, Beckers HJ. Corneal endothelial cell loss after Baerveldt glaucoma drainage device implantation in the anterior chamber. Acta
Ophthalmol. 2017 Feb;95(1):91-96.
21. Beatson B, Wang J, Boland MV, et al. Corneal edema and keratoplasty: risk factors in eyes with previous glaucoma drainage devices. Am J Ophthalmol. 2022; 238:27-35
22. Hill RA, Heuer DK, Baerveldt G, Minckler DS, Martone JF. Molteno implantation for glaucoma in young patients. Ophthalmology. 1991 Jul;98(7):1042-6.
23. Lloyd MA, Sedlak T, Heuer DK, Minckler DS, Baerveldt G, Lee MB, Martone JF. Clinical experience with the single-plate Molteno implant in complicated glaucomas. Update of a pilot study. Ophthalmology. 1992 May;99(5):679-87.
24. Pathak Ray V, Rao DP, Gulati I. Primary implantation of non-valved glaucoma-drainage-device in sulcus in iridocorneal endothelial syndrome. Int J Ophthalmol. 2019;12(11):1809-1811.
25. Freedman J, Iserovich P. Pro-Inflammatory Cytokines in Glaucomatous Aqueous and Encysted Molteno Implant Blebs and Their Relationship to Pressure. Investigative Ophthalmology & Visual Science 2013;54:4851-55.
26. Ishida K, Netland PA. Ahmed Glaucoma Valve implantation in African American and white patients. Arch Ophthalmol. 2006;124(6):800-6.
27. Abe RY, Tavares CM, Schimiti RB, Vasconcellos JP, VP. C. Ahmed Glaucoma Valve implantation for refractory glaucoma in a tertiary hospital in Brazil. Journal of Ophthalmology. 2015.
28. Won HJ, Sung KR. Hypertensive Phase Following Silicone Plate Ahmed Glaucoma Valve Implantation. J Glaucoma. 2016;25(4):e313-7.
29. Elhefney E, Mokbel T, Abou Samra W, Kishk H, Mohsen T, El-Kannishy A. Long-term results of Ahmed glaucoma valve implantation in Egyptian population. Int J Ophthalmol. 2018;11(3):416-21.
30. Dubey S, Sharma DK, Bhoot M, Pegu J, Gandhi M. Hypertensive Phase Following Silicone Plate Ahmed Glaucoma Valve Implantation. Journal of Glaucoma. 2017;26(3):e124.
31. Law SK, Kornmann HL, Giaconi JA, Kwong A, Tran E, Caprioli J. Early Aqueous Suppressant Therapy on Hypertensive Phase Following Glaucoma Drainage Device Procedure: A Randomized Prospective Trial. J Glaucoma.
2016;25(3):248-57.


 FP0436 : Primary implantation of Glaucoma Drainage Device in Secondary Glaucoma: comparison of AADI vs AGV
FP0436 : Primary implantation of Glaucoma Drainage Device in Secondary Glaucoma: comparison of AADI vs AGV
Leave a Comment