Dr. HAIMANTI CHOUDHURY
Dr.Hirendra Kumar Choudhury, Dr. HIMADRI CHOUDHURY
Abstract
Purpose: To evaluate the role of novel AI derived dry eye analyzer in evaluation of patients with ocular surface disorder (OSD).
Method: Prospective study to evaluate consecutive patients with OSD. They were subjected to Dry Eye work up by Dry Eye Analyzer including Non-invasive breakup time (NIBUT), Non-invasive Tear meniscus height, evaluation of meibomian gland function by built in infrared lighting & lipid layer thickness. Schirmer Test & ocular surface staining were also done for correlation.
Results: 250 patients with mean age 36.4±12.3 years were examined. According to the Schirmer test severe dry eye was found in 11.6%. NIBUT revealed dry eye in 18.8%. Meibomian gland disorder was found in 56%. Abnormal lipid layer thickness was found in 51.6%. Abnormal tear meniscus height was found in 9.4%. Correlation coefficient between Schirmer & NIBUT was 0.7.
Conclusion: Dry Eye Analyzer can diagnose cases hithero missed by conventional tests & has the advantage of being non invasive.
Full Text
AUTHORS
Dr Haimanti Choudhury, Dr Hirendra Kumar Choudhury, Dr Himadri Choudhury
ABSTRACT
Purpose: To evaluate the role of novel AI derived dry eye analyzer in evaluation of patients with ocular surface disorder (OSD).
Method: Prospective study to evaluate consecutive patients with OSD. They were subjected to Dry Eye work up by Dry Eye Analyzer including Non-invasive breakup time (NIBUT), Non-invasive Tear meniscus height, evaluation of meibomian gland function by built in infrared lighting & lipid layer thickness. Schirmer Test & ocular surface staining were also done for correlation.
Results: 250 patients with mean age 36.4±12.3 years were examined. According to the Schirmer test severe dry eye was found in 11.6%. NIBUT revealed dry eye in 18.8%. Meibomian gland disorder was found in 56%. Abnormal lipid layer thickness was found in 51.6%. Abnormal tear meniscus height was found in 9.4%. Correlation coefficient between Schirmer & NIBUT was 0.7.
Conclusion: Dry Eye Analyzer can diagnose cases hithero missed by conventional tests & has the advantage of being non invasive.
Introduction
Dry eye disease (DED) is a frequent cause of ocular irritation for which patients seek ophthalmic care. Due to wide variety of presenting symptoms, it is often unrecognised. We come across many patients with ocular surface discomfort for whom empirical dry eye therapy is administrated most of the time without confirming whether the symptoms are attributable to a true dry eye condition.
The ocular surface consists of conjunctival mucosa that lines the bulbar and palpebral surfaces, the corneal scleral limbus, the corneal epithelium and tearfilm. These components work physiologically along with eyelids and lacrimal functional unit (LFU) to maintain a healthy ocular surface and form an “ocular surface system” Dry eye is a common disorder of the preocular tear film that results in damage to the ocular surface and is associated with symptoms of ocular discomfort. It is defined as a multifactorial disease of tears and ocular surface results in symptoms of discomfort, visual disturbance and tear film instability with potential damage of the ocular surface. It is accompanied by increased osmolarity of the tear film and inflammation of the ocular surface.1
Though the definition of the disease is seemingly clear, the diagnosis presents multiple challenges to the clinician. First of all, no gold standard tests for diagnosis exists and no single test is sufficient for diagnosis due to poor reliability, multiple causes of the disease and lack of well-defined cut-off values to distinguish disease from normal.2 Secondly, the signs and symptoms do not always correlate and both can vary based on influences, such as diurnal or seasonal fluctuations.3 In addition, many of the tests are invasive in nature and this may influence the outcome. Lastly, other conditions can mimic dry eye such as ocular allergy. Due to these challenges, alternatives to traditional dry eye testing have emerged.4
Many tests used in the diagnosis of DED rely on an experienced observer for image interpretation, which may be considered subjective and can result in diagnostic dilemma. Since artificial intelligence (AI) systems are capable of advanced problem solving, use of such techniques could lead to more objective diagnosis. Recent success in application of AI to medicine is mainly due to advancements in the sub-field of machine learning, which has enabled to automatically classify images and predict medical outcomes. Powerful machine learning techniques have been utilised to understand nuances in patient data and medical images, aiming for consistent diagnosis and stratification of disease severity.5
The purpose of this study is to evaluate the role of novel AI derived dry eye analyzer in evaluation of patients with ocular surface disorder (OSD).
METHODS
A prospective study was conducted to evaluate consecutive patients with OSD. The study was approved by the Ethics Committee of our hospital and was performed in accordance with the guidelines of the Declaration of Helsinki. Informed consent was obtained from the study subjects at the time of enrolment. A total of 250 consecutive patients attending ophthalmology OPD with ocular surface symptoms were included.
Exclusion criteria –
Post operative patients
Patients suffering from acute ocular infections, extensive corneal or conjunctival pathology.
Chronic dacryocystitis
Patients who were on topical lubricants for the last six months.
Before tests some questions were asked to the patients regarding various symptoms they were suffering. A detailed history taking was done followed by slit-lamp biomicroscopic examination They were subjected to Dry Eye work up by Dry Eye Analyzer including Non-invasive breakup time (NIBUT), Non-invasive Tear meniscus height, evaluation of meibomian gland function by built in infrared lighting & lipid layer thickness. Schirmer Test & ocular surface staining were also done for correlation.
Non invasive break up time
We used the novel AI derived dry eye analyzer (Dry eye diagnostic system, Mediworks) to assess the NIBUT. It uses Placido ring projection system with visible light to do NIBUT examination. The examination scope is up to 8mm corneal diameter which brings much more comprehensive diagnostic outcome. It notes image distortion and time from opening the eyes to the first sign of image distortion. After taking one video, it automatically acquires the first break up time, average break up time, break up distribution, break up area, percentage curve and time distribution.
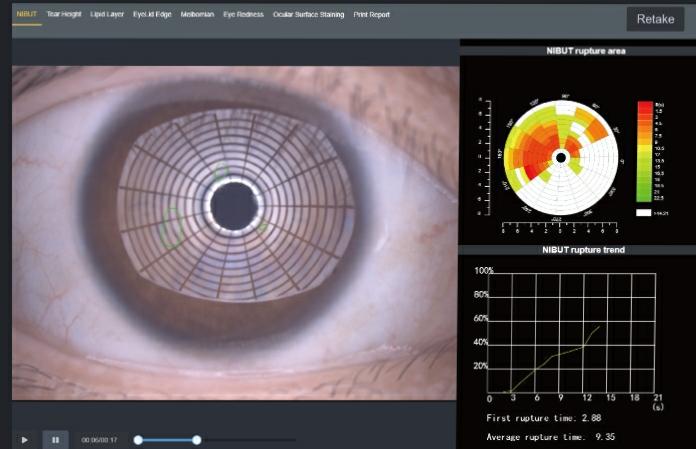
Fig:1 NIBUT – Dry eye
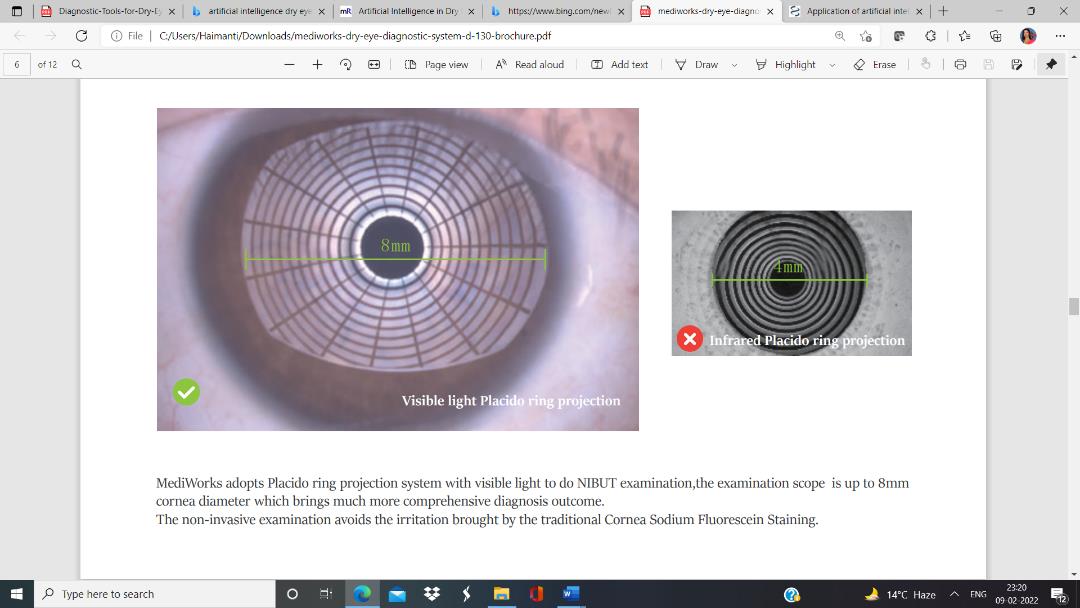
Fig:2 NIBUT – Normal eye
Grade 0 Normal,First Rupture Time: 10 s Average Rupture Time: 14 s
Grade 1 Warning, First Rupture Time: 6-9 s Average Rupture Time: 7-13 s
Grade 2 Dry eye, First Rupture Time: 5 s Average Rupture Time: 7 s
The normal time for TBUT is 15-20 seconds and TBUT with values less than10 seconds suggest an abnormal tear film, with values 5-10 seconds considered marginal and less than 5 seconds indicative of dry eye.6
Non-invasive Tear meniscus height
A video is taken and AI identification system depicts Tear Meniscus area and measures the tear height automatically with a built in ruler. It is measured in line with the pupil centre. It evaluates tear secretion amount and continuity objectively. Normal tear meniscus height is ≥0.2mm.
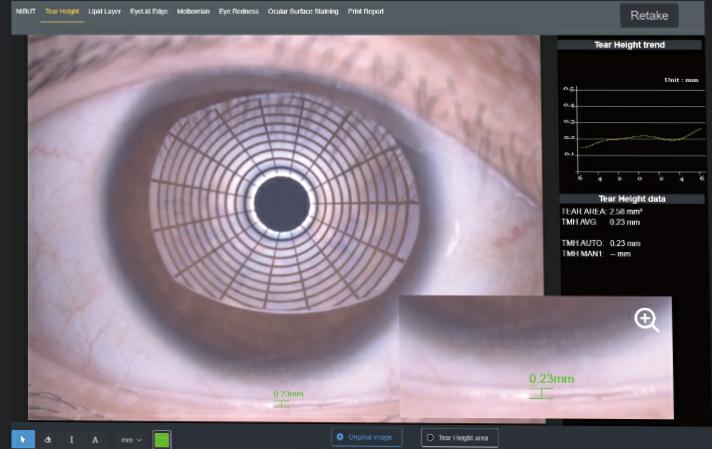
Fig:3 Tear meniscus height 0.23
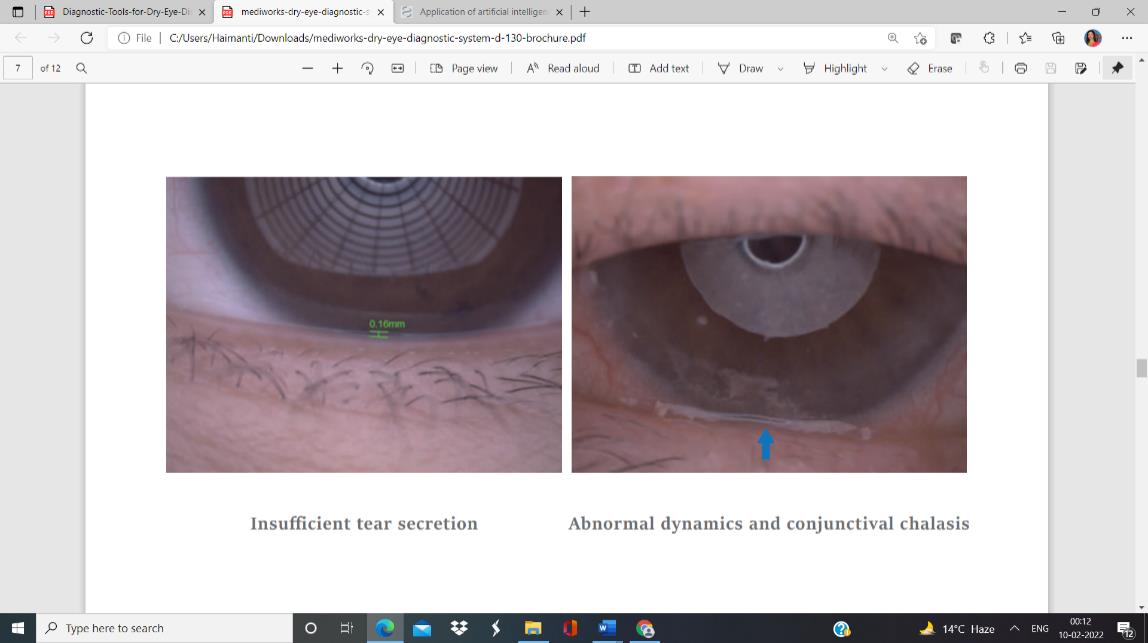
Fig:4 Tear meniscus height 0.16
Meibomian gland function
Meibomian gland disease is a major, and perhaps the most common, etiologic factor in the pathogenesis of many subtypes of DED. Clinical diagnosis is often limited to examination of the lid margin by slit lamp to assess the degree of inspissation and telangiectasias, as well as subjective assessment of meibomian gland openings and meibum quality. techniques. Recently, infrared based non-contact imaging modalities of meibomian gland have offered detailed imaging to guide the diagnosis and treatment of MGD-related DED. Unique Built-in infrared lighting system provides a larger scope of capturing Meibomian gland image of both upper and lower lids.
Meibomian gland dropout as assessed by this method correlates well with signs and symptoms of dry eye disease. These imaging modalities can provide valuable objective information about the integrity of the glands. Adjustable depth of field makes the glands more prominent and distinguishable against the background.
Grade 0: No Meibomian Glands Loss
Grade 1: Meibomian Glands Loss < 1/3
Grade 2: Meibomian Glands Loss 1/3-2/3
Grade 3: Meibomian Glands Loss >2/3
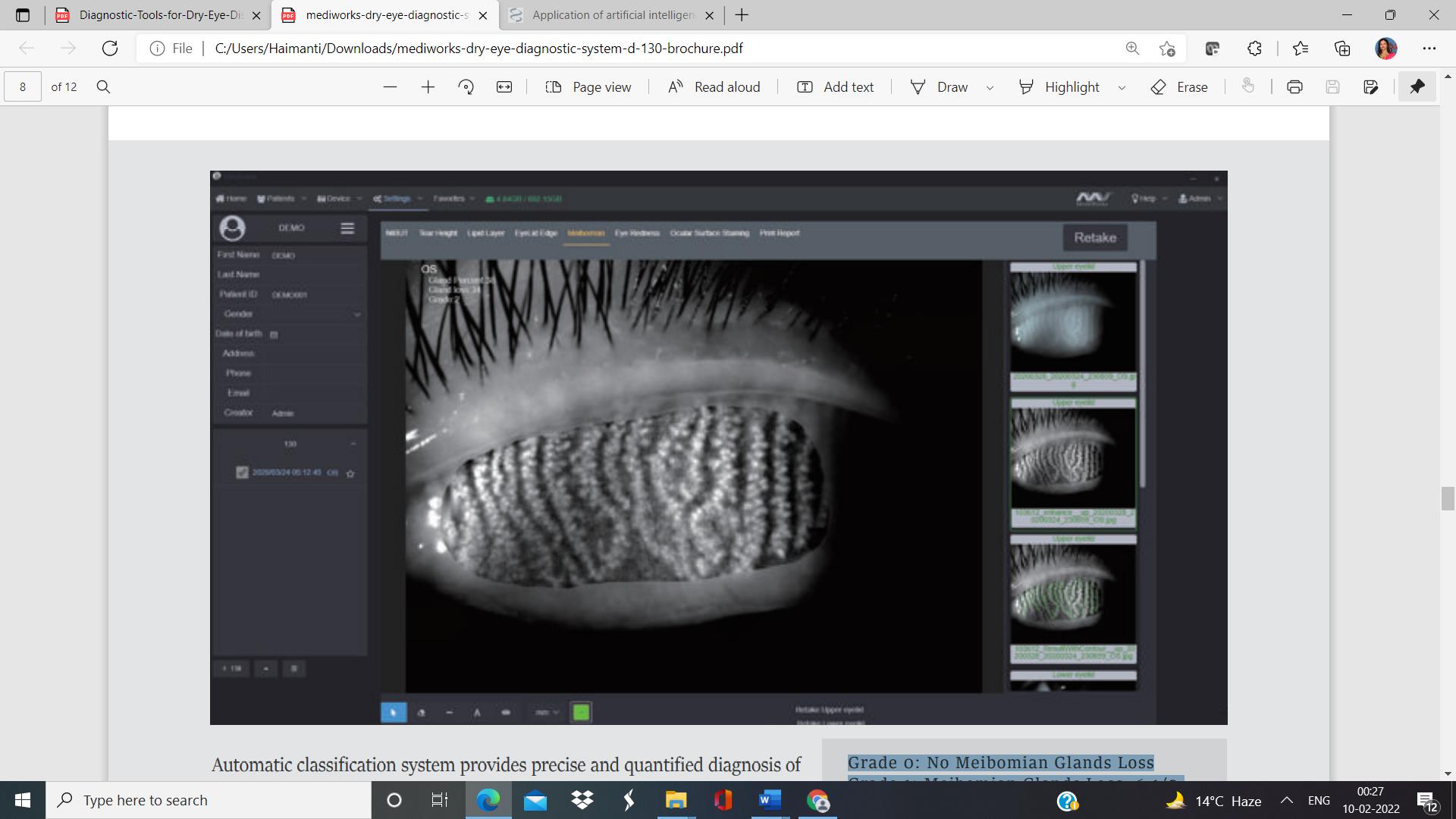
Fig 5: No Meibomian gland loss
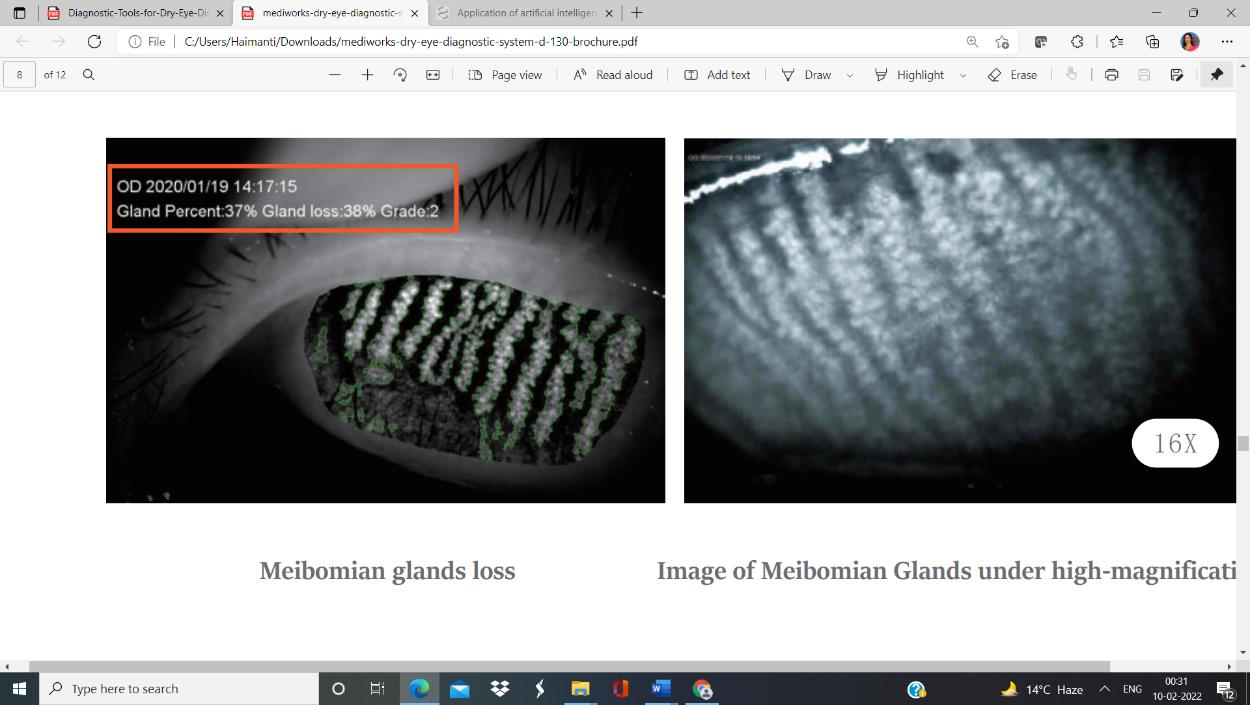
Fig 6: Grade 2 Meibimian gland loss
Lipid layer thickness
The principle of white light interferometry is used to get a quantitative value of lipid layer thickness. The patient is instructed to blink while a recording is taken. Thicker lipid layers are indicated by the bronze reflections with lot of colourful fringes. White colourless reflections are observed in patients with thin lipid layer.
Grade 1: <30
Grade 2: 30-60
Grade 3: 60-80
Grade 4: >80
(Unit:nm)
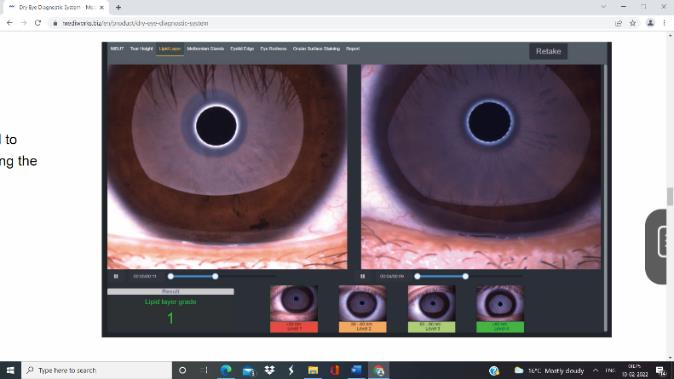
Fig 7: Normal lipid layer (left) , thin lipid layer (right)
Schirmer test
It is the most commonly used test for the assessment of tear secretion. It measures total tear secretions. It is performed with help of a 5×35 mm strip of Whatman-41 filter paper which is folded 5 mm from one end and kept in the lower fornix at the junction of lateral one-third and medial two-thirds. The patient should close his/her eyes upto 5 minutes. After 5 minutes wetting of the filter paper strip from the bent end is measured. Normal values of Schirmer-1(without anaesthetic) test are more than 15 mm. Values of 5-10 mm are suggestive of moderate to mild dry eye and less than 5 mm of severe dry eye.
Ocular surface dye staining
Use of dyes can help assess the superficial ocular surface to detect damage that is often present in DED. We used fluorescein and lissamine green. Fluorescein dye is taken up by corneal and conjunctival tissue where there is disruption in the intercellular junctions. The staining of the cornea is seen with use of the cobalt blue filter on the slit lamp whereas the conjunctival staining is seen with a yellow (blue-free) filter. The classical staining of the cornea in dry eye includes superficial punctate keratitis concentrated in the intrapalpebral or inferior area initially. Lissamine green evaluates the conjunctival surface by staining areas not properly covered by mucin. The Oxford grading scale was used.7
RESULTS
250 patients with symptoms of OSD were examined. Mean age was 36.4±12.3 years. There were 167 females (66.8%) and 83 males (33.2%). According to the Schirmer test, severe dry eye was found in 11.6% patients and mild to moderate dry eye was found in 39.2%. NIBUT revealed dry eye in 18.8% patients. Meibomian gland disorder was found in 56%. Abnormal lipid layer thickness was found in 51.6%. Abnormal tear meniscus height (TMH) was found in 9.4%. Ocular surface staining was found positive in 12.8% patients. Correlation coefficient between Schirmer & NIBUT was 0.7. 23.2% patients who had complaints of OSD showed negative results in all parameters of testing. 76.8% were diagnosed to have dry eye in one or more of the parameters. Only 9.2% patients tested positive in all the parameters. 14.4% patients who had normal values in Schirmer test and showed normal staining were diagnosed to have some form of tear film deficiency when tested by the AI derived Dry eye analyzer.

Fig 8: Percentage of patients having dry eye Vs no dry eye

Fig 9: Percentage of patients with various tear component deficiency
DISCUSSION
AI application in conjunctiva and tear film mainly focus on the diagnosis of dry eye. The diagnosis of dry eye is complicated and there is not a single perfect reference standard of it.8 Instead, a variety of examinations can provide supportive information. DED is one of the most common eye diseases worldwide, with a prevalence of between 5 and 50%, depending on the diagnostic criteria used and study population.9
DED is divided into two subtypes defined by the underlying mechanism of the disease: (i) aqueous deficient DED, where tear production from the lacrimal gland is insufficient and (ii)evaporative DED (the most common form), which is typically caused by dysfunctional meibomian glands in the eyelids.
Artificial intelligence (AI) was defined in 1955 as “the science and engineering of making intelligent machines”, where intelligence is the “ability to achieve goals in a wide range of environments” Mccarthy2006 AI proposal. 10
In our study, 23.2% patients who had complaints of OSD showed negative results in all parameters of testing and DED was ruled out in them. 9.2 % patients were considered to be having severe DED when all the diagnostic tests such as NIBUT, Non-invasive Tear meniscus height, evaluation of meibomian gland function, lipid layer thickness, Schirmer Test & ocular surface staining showed positive results. Our study reported high positivity of Meibomian gland disorder (56%) and Abnormal lipid layer thickness (51.6%) which is otherwise missed by conventional testing methods.
CONCLUSION
Novel artificial intelligence derived Dry Eye Analyzer can diagnose cases hithero missed by conventional tests & has the advantage of being non invasive. It has the potential of replacing the conventional invasive dry eye tests in the present times when both patients and clinicians want to maintain safe distance and prioritize on sanitization methods.
REFERENCES
1. The definition and classification of dry eye disease: report of the Definition and Classification Subcommittee of the International Dry Eye WorkShop (2007), Ocul Surf, 2007;5:75-92.
2. Savini G, Prabhawasat P, Kojima T, et al., The challenge of dry eye diagnosis, Clin Ophthalmol, 2008;2:31–55
3. McGinnigle S, Naroo SA, Eperjesi F, Evaluation of dry eye, Surv Ophthalmol, 2012;57:293–316
4. Methodologies to diagnose and monitor dry eye disease: report of the Diagnostic Methodology Subcommittee of the International Dry Eye WorkShop (2007), Ocul Surf, 2007;5:108–52.
5. Artificial Intelligence in Dry Eye Disease
Andrea M. Storås, Inga Strümke, Michael A. Riegler, Jakob Grauslund, Hugo L. Hammer, Anis Yazidi, Pål Halvorsen, Kjell G. Gundersen, Tor P. Utheim, Catherine Jackson
medRxiv 2021.09.02.21263021; doi: https://doi.org/10.1101/2021.09.02.21263021
Now published in The Ocular Surface doi: 10.1016/j.jtos.2021.11.004
6. Pflugfelder SC, Tseng SC, Sanabria O, Kell H, Garcia CG, Felix C, Feuer W, Reis BL. Evaluation of subjective assessments and objective diagnostic tests for diagnosing tear-film disorders known to cause ocular irritation. Cornea. 1998 Jan;17(1):38-56. doi: 10.1097/00003226-199801000-00007. PMID: 9436879.
7. Bron AJ, Evans, VE, Smith JA, Grading of corneal and conjunctival staining in the context of other dry eye tests, Cornea, 2003;22:640-50
8. Andrea M. Storås, Inga Strümke, Michael A. Riegler, Jakob Grauslund, Hugo L. Hammer, Anis Yazidi, Pål Halvorsen, Kjell G. Gundersen, Tor P. Utheim, Catherine J. Jackson,
Artificial intelligence in dry eye disease,The Ocular Surface,Volume 23,2022,Pages 74-86,ISSN 1542-0124,https://doi.org/10.1016/j.jtos.2021.11.004.
9. Wu X, Liu L, Zhao L, et al. Application of artificial intelligence in anterior segment ophthalmic diseases: diversity and standardization. Ann Transl Med. 2020;8(11):714. doi:10.21037/atm-20-976
10. ARVO Annual Meeting Abstract | July 2018
Diagnosis of dry eye subtype by artificial intelligence software based on the interferometric fringe pattern of the tear film obtained with the Kowa DR-1α instrument
Reiko Arita; Katsumi Yabusaki; Takanori Yamauchi; Tadashi Ichihashi; Naoyuki Morishige
Investigative Ophthalmology & Visual Science July 2018, Vol.59, 1965. doi:



 FP2323 : Novel artificial intelligence (AI) derived dry eye analyzer – is it here to stay?
FP2323 : Novel artificial intelligence (AI) derived dry eye analyzer – is it here to stay?
Leave a Comment