DR AVADHESH OLI
DR. (COL) SHRIKANT WAIKAR, Dr.Dattakiran Joshi
Abstract
Psuedophakic cystoid macular edema (PCME) mostly responds to topical drops, however intra-vitreal injections may be required. Suprachoroidal injection of preservative free Triacinolone acetonide (SCTA) has been used recently for macular edema of uveitis and diabetic retinopathy. We studied safety and efficacy of SCTA in PCME.
Methods
20 patients with PCME not responding to topical medication were enrolled. The drug was administered with an innovated 26 G needle with intracath sleeve. Change in BCVA, macular thickness were primary outcome measures.
Results
The baseline visual acuity was logMAR 0.525 which improved to log MAR 0.06 at 1 month (p<0.00) The baseline CMT reduced from 451 micron to 261 micron (p<0.00). No significant complications like raised IOP, RD were noted. Conclusion This modified method of SCTA injection is cost effective and safe for treatment of PCME. The technique has potential of wider applicability in countries where cost of treatment is a concern.
Full Text
Running title: Safety and efficacy of suprachoroidal triamcinolone for psuedophakic CME
Psuedophakic Cystoid macular oedema (PCME) mostly responds to topical steroids and NSAIDs, however intra-vitreal injections may be required for nonresponsive cases. Suprachoroidal injection of preservative free Triamcinolone acetonide (SCTA) is a recent addition to for treatment of cystoid macular edema of uveitis and diabetic retinopathy. We studied safety and efficacy of SCTA in psuedophakic CME.
Methods:
20 patients with PCME not responding to topical medication were enrolled. Patients with other macular pathology like DME were excluded. The drug was administered with an innovated 26 G needle with intracath sleeve in OT. Change in BCVA, macular thickness and complications, if any at 6 months were primary outcome measures.
Results:
The baseline visual acuity was logMAR 0.525 which improved to log MAR 0.06 at 1 month (p<0.00) The baseline CME reduced from 451 micron to 261 micron at 2 months (p<0.00). No significant complications like raised IOP, RD or uveitis were noted.
Conclusions:
This modified method of SCTA injection is cost effective and safe for treatment of PCME. This technique may be concurrently used with cataract surgery in patients with high risk for developing macular edema like diabetic retinopathy or uveitic cataracts.
Key words:
Macular edema, Suprachoroidal steroids, Triamcinolone, Cataract surgery complications
Introduction
Patient expectations have increased multifold after the cataract surgery due to the advancement in surgeon skills, operating machines and premium intraocular lenses. The modern-day cataract surgery has become like a refractive surgery. Pseudophakic Cystoid macular edema (PCME) is a common cause of reduced vision, after cataract surgery, and the incidence may vary from 0.5 – 2.4%.[1] The incidence of PCME is more in patients with diabetes and uveitis. The cost of treatment of CME adds on to the financial burden and is more significant in economically underprivileged population. The patients who develop PCME end up paying more for their medical expenses.[2] The cellular pathological cause of macular edema is multifactorial nevertheless inflammation plays a key role.
Topical steroids and nonsteroidal anti-inflammatory (NSAID) drops are the mainstays of therapy for the initial postoperative period; however, chronic PCME requires periocular or intravitreal injections which might cause significant side effects like endophthalmitis, retinal detachment, cataract and glaucoma.[3] Steroid implants, are sustained released drug-eluting devices, which have recently become popular. Although the steroid implants are costly, however, they have lesser side effects like glaucoma and longer duration of action.
Recent studies on outcomes of depot steroid injection of triamcinolone into suprachoroidal space have shown promising results in terms of improved efficacy, reduced side effects and longer duration of action in patients with macular edema due to uveitis and retinal vein occlusion.[4-6] The micro-needle used for injection is short and does not enter the vitreous cavity, which might reduce the chances of damage to ocular tissues. Although, the procedure can be performed in an office setting under sterile precautions, but the critical limitation is the high cost and non-availability of the specialized microneedle.
Recently innovative technique of delivery of SCTA using intracath and 26 G needle has been reported which seems quiet practical and feasible. The Superior efficacy of SCTA could be attributed to relative posterior diffusion of the drug and sustained therapeutic levels in the vitreous cavity. The current study was undertaken to study the safety and efficacy of SCTA delivered with such modified technique in patients with PCME after cataract surgery who failed to respond to topical drops.
Methods
Institutional ethical committee clearance was taken for the study from the institution. Patients with chronic PCME (PCME persisting despite of three months of topical NSAIDs- 1% Nepafenac drops, Alcon India) with macular thickness > 300 micron on optical coherence tomography (OCT) were enrolled after informed consent. Patients with hypersensitivity to TA, immune deficiency, glaucoma, uveitis, diabetic retinopathy, and other retinal vascular diseases were excluded from the study.
The comprehensive ophthalmological examination including visual acuity, slit lamp biomicroscopy detailed retina evaluation and optical coherence tomography (OCT) was done as per the study protocol.
Preparation of the Injection
The outer sheath of a 22 G intracath (25 mm length X 0.9 mm outer diameter (Anina India)) was cut into two equal halves and 26 G needle (length 13 mm) was passed through the lumen. Final measurement was taken with calipers and care was taken to exposure terminal 1 mm beveled end of the 26 G needle. Preservative free Triamcinolone 4mg/.01 ml (Auro Lab India) was taken in a 1 CC syringe and the 26G needle was connected to the syringe.
One intracath sheath (length of 12 mm) has adequate length to make two separate injections. (Fig 1 A)
The periocular area was cleaned and draped after instilling Proparacaine drops and Betadine 5% with a contact period of 5 minutes. Lid speculum was applied and patient was asked to look in temporal direction exposing the superonasal quadrant. A point was marked 4 mm from limbus and the sclera was pierced keeping the needle perpendicular and the beveled tip pointing away from the limbus. Gentle pressure was applied to create dimple on the sclera and the drug was injected gently.
The needle tip was withdrawn and cotton tip or sponge was used to avoid backflow of the drug. Any procedure related complications were documented. Patients were followed up on day 1, 7, 30 and 90 after the injection. At every follow-up visit visual acuity, intraocular pressure, central macular thickness (CMT) and complications if any, were documented. (Fig 1 B)
Results
This study included 20 patients with chronic PCME despite of topical NSAID drops for three months. All the patients had undergone uncomplicated cataract surgery with single piece intraocular lens in the bag implantation using phacoemulsification.
Out of twenty patients 12 (60%) were males and 8 (40%) were females. The mean age of the patients was 54.35 years (SD 7.7 years). The mean duration of treatment of PCME with topical NSAID was 13.75 weeks (SD 1.25) with a range of 12 – 16 weeks.
The table 1 depicts demographic and clinical data of 20 patients who received 0.1 mg Triamcinolone through the suprachoroidal route for psuedophakic CME
The mean baseline best corrected visual acuity in this series of twenty patients was 0.52 log MAR (Snellen equivalent 6/24) which improved to 0.06 log MAR (Snellen equivalent 6/6) at the end of four weeks (p<0.005). The central macular thickness reduced from a mean of 451.90 microns at baseline to 261 microns (p<0.005). The intraocular pressure at the baseline was 13.30 mmHg which remained within normal limits at 13.50 mmHg(p=0.64). No significant side effects like uveitis, endophthalmitis, retinal detachment or glaucoma were noted however 2 patients complained of moderate eye pain which subsided with NSAID tablets. Five patients had localised subconjunctival haemorrhage which resolved within one week. At the end of three months, the visual acuity was maintained.
Discussion
PCME is an important complication after the cataract surgery and it is managed using topical NSAIDs, along with steroids; however, intravitreal anti-VEGF or steroid injections may be required for cases not responding to topical drops. The alternative treatment is the use of intravitreal steroid implants due to less effect on IOP, nonetheless the cost is astronomically high and other complications like retinal detachment and endophthalmitis are reported. Quest for finding newer ocular drug formulations and delivery routes have led to description of newer techniques. A recent phase 3 trial on uveitis has demonstrated the efficacy of supra choroidal triamcinolone SCT in macular edema due to retinal vein occlusion & uveitis.[4,6,7]
In preliminary studies, SCT was found to reach a high concentration in the posterior segment with lesser side effects like glaucoma and cataract. The therapeutic level of the drug in the posterior segment was reported to be ten times higher than the anterior segment.[5] SCT is localised in the suprachoroidal space, at the site of pathology and the local concentration remains high, which improves efficacy and prolongs the duration of action due to depot formulation.
Currently, the limitation for the broader applicability of SCT is non-availability of an appropriate needle for safe injection procedure. In this study we used a low-cost alternative injection system which was easily available & designed from inexpensive sterile material available in the operating room. Use of one intracath to prepare two injections made the procedure further economical. The outer sheath of intracath acts as a guard to the sharp end of the needle which pierces the sclera and remains in suprachoroidal space. The SCT localizes in the suprachoroidal space, which is a potential space and opens up while injecting the drug. In cadaver experiments the capacity of suprachoroidal space was found to be 1 ml so the smaller volume of 0.1 ml injected in various clinical studies should not be a cause of concern.
While injecting the drug it should be ensured that terminal 1 mm of the needle tip should be exposed otherwise if the bevelled end is also covered the drug might leak from the gap between the intracath and needle. Another important step is to apply gentle pressure after the sclera is pierced to create a dimple to ensure adequate penetration of the needle so that the drug is injected into the suprachoroidal space.
(Fig 1 B) The bevel of the needle should be pointing away from limbus so that the drug diffuses posteriorly.
The SCTA is practically extraocular procedure like the posterior sub-tenon triamcinolone injection, but direct visualization of the needle makes SCTA safer and more predictable. In addition, since the vitreous cavity is not entered, the chances of sight threatening complications like endophthalmitis, retinal tear, retinal detachment, lens injury are fewer.
Two patients reported moderate eye pain immediately after the procedure, which subsided after one hour with NSAID tablets. The expansion of suprachoroidal space with SCT could be the cause of the pain. Mild conjunctival congestion at the site of injection was noted in 5 patients, which resolved within one week.
SCTA has sustained therapeutic effect as the drug is a depot formulation as supported by pharmacokinetic studies. SCTA may aid in stabilizing the vision in the long-term. [8] As the frequency of the injections required would be less, it would also drastically cut down the cost of repeated injections. Compared to expensive alternatives, the SCTA injected with this innovative technique would benefit the patients from weaker sections of the society.
The patients in this study had normal IOP at baseline which remained within the normal range at the end of the study. None of the patients required to use of any anti-glaucoma medication. Due care should be exercised while using SCTA in patients with glaucoma and frequent IOP checks should be done in the follow-up period. The efficacy of SCTA has been well established in randomized trials conducted in patients with CME following uveitis. We also excluded patients with other causes of macular edema like patients with diabetes and uveitis, which may not be practical in a real-world scenario.
The results of current study are encouraging in terms of safety and efficacy of SCTA in psuedophakic CME. The use of innovative technique for injection procedure makes the procedure economical as well as affordable and has potential to become a technique for the masses in macular edema of varied etiology. Larger randomized controlled trials might be undertaken to further establish the safety and efficacy of SCTA.
Authors do not have any financial disclosures to make, the study was not funded by any grants.
Figure Legends
Fig 1 A The outer sheath of intracath is cut and used to cover the 26-gauge needle till the bevelled edge 1 B Optical coherence tomography showing TA deposited in suprachoroidal space (Upper pane) and completely resolved CME (Lower pane)
References
1. Hoffman RS, Braga-Mele R, Donaldson K, et al. Cataract surgery and nonsteroidal antiinflammatory drugs. Journal of cataract and refractive surgery. 2016;42(9):1368-1379.
2. Schmier JK, Covert DW, Hulme-Lowe CK, Mullins A, Mahlis EM. Treatment costs of cystoid macular edema among patients following cataract surgery. Clinical ophthalmology (Auckland, NZ). 2016;10:477-483.
3. Han JV, Patel DV, Squirrell D, McGhee CN. Cystoid macular oedema following cataract surgery: A review. Clinical & experimental ophthalmology. 2019;47(3):346-356.
4. Campochiaro PA, Wykoff CC, Brown DM, et al. Suprachoroidal Triamcinolone Acetonide for Retinal Vein Occl{Citation}usion: Results of the Tanzanite Study. Ophthalmology Retina. 2018;2(4):320-328.
5. Gilger BC, Abarca EM, Salmon JH, Patel S. Treatment of acute posterior uveitis in a porcine model by injection of triamcinolone acetonide into the suprachoroidal space using microneedles. Investigative ophthalmology & visual science. 2013;54(4):2483-2492.
6. Yeh S, Kurup SK, Wang RC, et al. SUPRACHOROIDAL INJECTION OF TRIAMCINOLONE ACETONIDE, CLS-TA, FOR MACULAR EDEMA DUE TO NONINFECTIOUS UVEITIS: A Randomized, Phase 2 Study (DOGWOOD). Retina. 2019;39(10):1880-1888.
7. Wykoff CC, Khurana RN, Lampen SIR, et al. Suprachoroidal Triamcinolone Acetonide for Diabetic Macular Edema: The HULK Trial. Ophthalmology Retina. 2018;2(8):874-877.
8. Chen M, Li X, Liu J, Han Y, Cheng L. Safety and pharmacodynamics of suprachoroidal injection of triamcinolone acetonide as a controlled ocular drug release model. Journal of controlled release : official journal of the Controlled Release Society. 2015;203:109-117.
Table 1 Demographic and clinical data
| SEX | AGE | NSAID | BCVA PRE | BCVA POST | CMT PRE | CMT POST | IOP PRE | IOP POST |
|---|---|---|---|---|---|---|---|---|
| Male | 55 | 14 | 0.6 | 0 | 473 | 287 | 12 | 15 |
| Male | 54 | 12 | 0.5 | 0 | 544 | 210 | 12 | 14 |
| Female | 67 | 14 | 0.4 | 0.1 | 345 | 270 | 14 | 12 |
| Male | 46 | 15 | 0.6 | 0 | 447 | 269 | 16 | 14 |
| Female | 43 | 16 | 0.5 | 0 | 437 | 266 | 13 | 12 |
| Female | 43 | 12 | 0.6 | 0.1 | 332 | 259 | 14 | 12 |
| Male | 55 | 13 | 0.5 | 0.2 | 433 | 271 | 12 | 14 |
| Female | 58 | 15 | 0.4 | 0.1 | 346 | 258 | 13 | 14 |
| Male | 62 | 14 | 0.5 | 0.2 | 632 | 267 | 15 | 14 |
| Male | 64 | 13 | 0.6 | 0 | 542 | 242 | 12 | 14 |
| Male | 55 | 14 | 0.6 | 0 | 456 | 287 | 12 | 15 |
| Male | 54 | 12 | 0.5 | 0 | 544 | 250 | 12 | 14 |
| Female | 66 | 14 | 0.5 | 0.1 | 345 | 250 | 14 | 12 |
| Male | 46 | 14 | 0.6 | 0 | 447 | 269 | 16 | 14 |
| Female | 43 | 16 | 0.5 | 0 | 437 | 266 | 13 | 12 |
| Female | 45 | 12 | 0.6 | 0.1 | 336 | 259 | 14 | 12 |
| Male | 55 | 13 | 0.5 | 0.2 | 432 | 273 | 12 | 14 |
| Female | 58 | 15 | 0.4 | 0 | 346 | 258 | 13 | 14 |
| Male | 62 | 14 | 0.5 | 0.1 | 632 | 267 | 15 | 14 |
| Male | 56 | 13 | 0.6 | 0 | 532 | 242 | 12 | 14 |
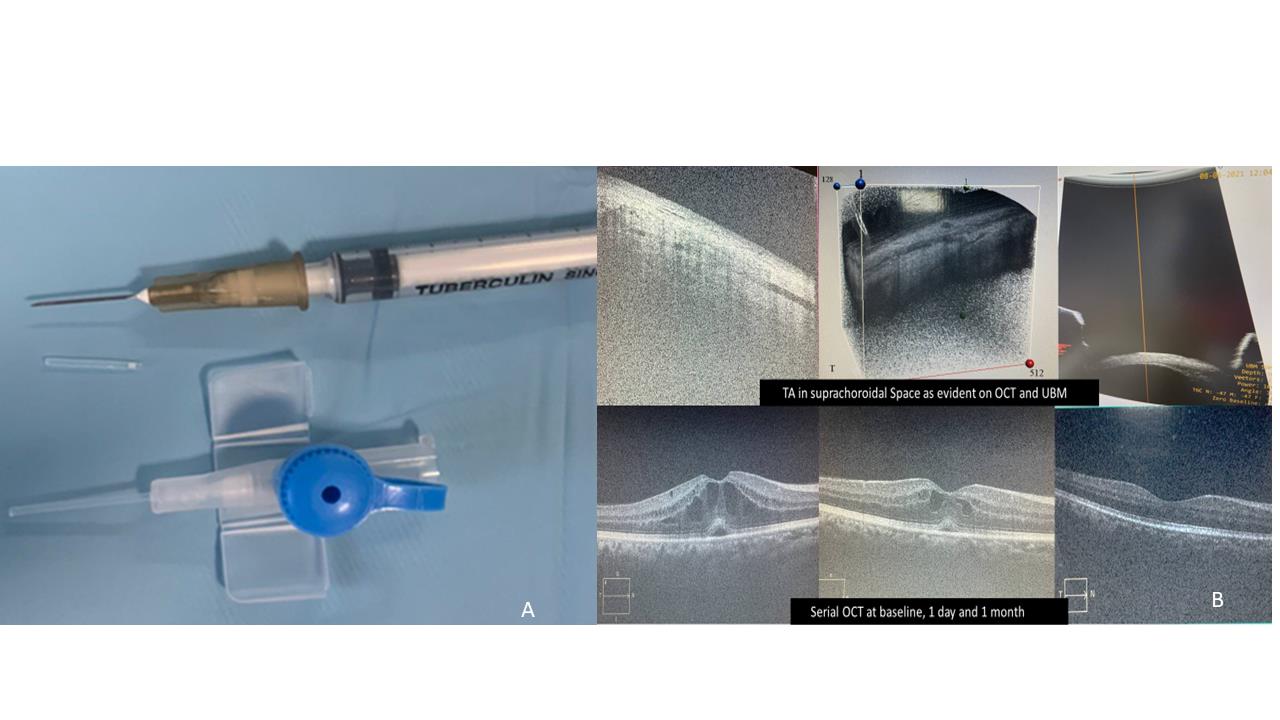
Fig 1 A The outer sheath of intracath is cut and used to cover the 26-gauge needle till the bevelled edge 1 B Optical coherence tomography showing TA deposited in suprachoroidal space (Upper pane) and completely resolved CME (Lower pane)


 FP0968 : Safety and efficacy of suprachoroidal triamcinolone for psuedophakic CME
FP0968 : Safety and efficacy of suprachoroidal triamcinolone for psuedophakic CME
Leave a Comment